Left: Cells expressing the photoactivatable GEF construct increase their forces exerted on the substrate when activated by light.
Cells are contractile organisms capable of migrating, changing their shape or dividing to suit their environment. In all these processes, the cells reorganize their overall structure or cytoskeleton in a matter of minutes. To increase their contractility, the cells increase the activity of the RhoA GTPase in the membrane by triggering its activator, the GEF molecule, locally.
The IBEC researchers have managed to direct the GEF molecule using light. To do this, they used a system of two proteins that bind with great affinity when exposed to a blue beam. One of the proteins in the pair binds to the membrane, and the other is anchored to the GEF activator. When the cell is illuminated with blue light, the GEF activator connects with the membrane protein, promoting cell contraction. “The presence of this GEF factor in the membrane is sufficient to increase the contractility of the illuminated cell up to 50%,” says Léo Valon, an IBEC researcher and the study’s lead author.
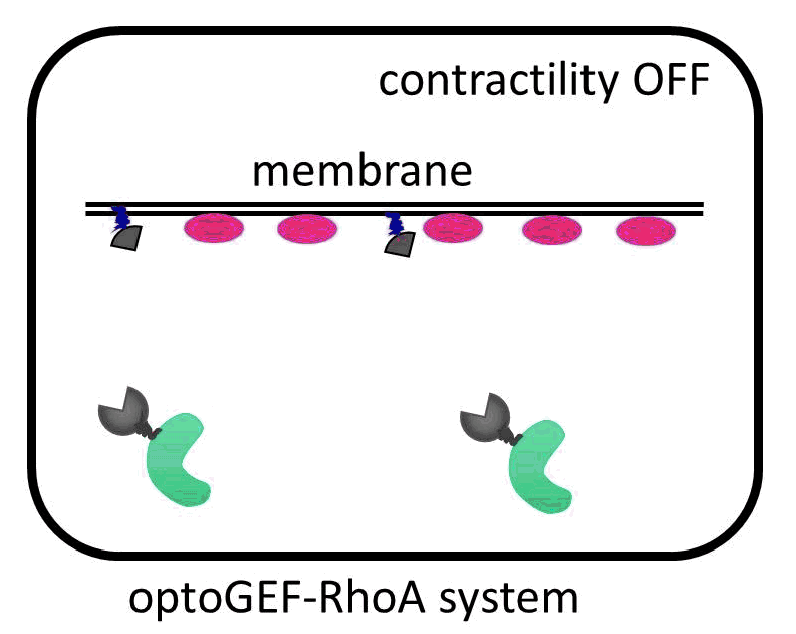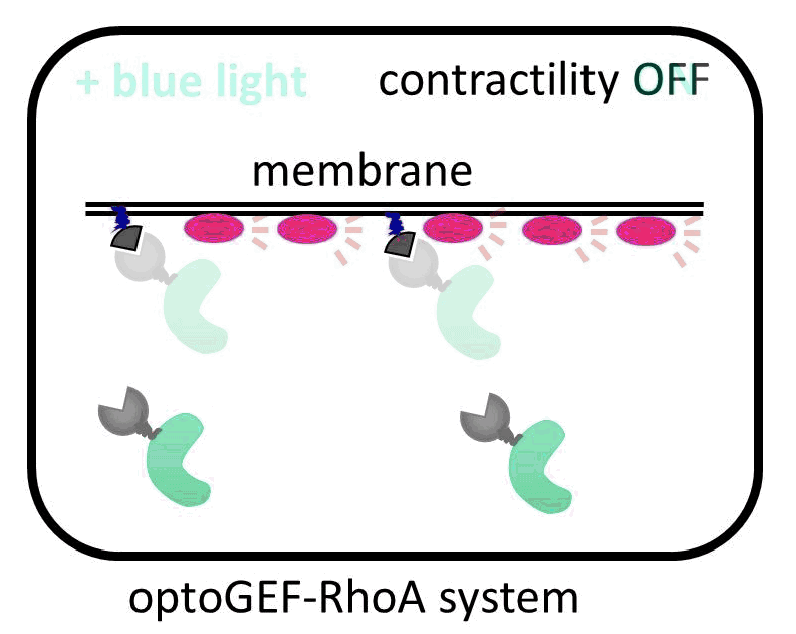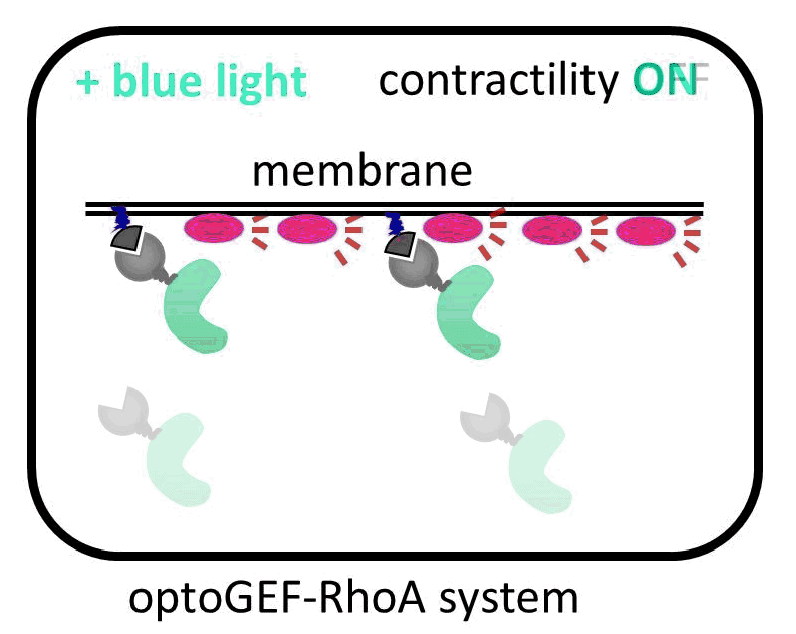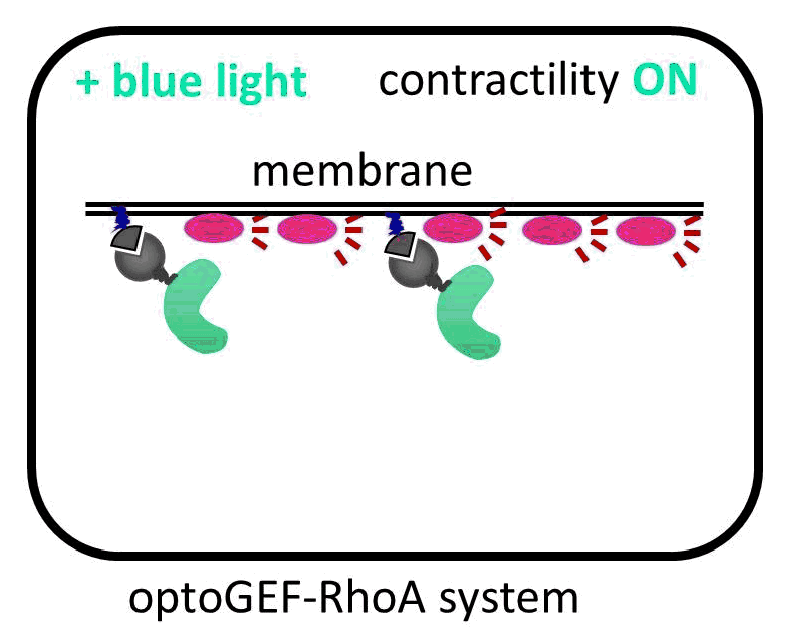 Activation of GEF contractility by turned on and off by light.
Activation of GEF contractility by turned on and off by light.
The increase in the forces exerted by the cells is completely reversible. “This allows us to generate local and periodic patterns of contraction and relaxation, simply by alternating the exposure of cells to light with periods of darkness,” continues Léo. The researchers were able to contract an epithelium in just five minutes using one pulse of light.
“The tools we have developed have many applications in mechanobiology,” says Xavier Trepat, ICREA researcher at IBEC and head of the group responsible for the research. “Being able to control the forces that make the cells will allow us to carry out detailed studies of phenomena such as cell adhesion and mechanotransduction at the subcellular level, and tissue deformation and remodeling at the supracellular level.”
In addition to the mechanobiology applications, the researchers predict that the new tool could be applied in the study of embryonic morphogenesis. “Because these optogenetic tools involve the use of equipment and genetic manipulations that are common in any laboratory, we expect them to become a benchmark technique for the control of cellular forces in different models,” says Xavier.
The study was made possible thanks to the financing of MINECO, the Generalitat de Catalunya and the ERC. The research team included Léo Valon, Ariadna Marín-Llauradó and Xavier Trepat from IBEC, as well as Thomas Wyatt and Guillaume Charras from the London Center for Nanotechnology.
—
Reference article:
Léo Valon, Ariadna Marín-Llauradó, Thomas Wyatt, Guillaume Charras & Xavier Trepat (2017). Optogenetic control of cellular forces and mechanotransduction. Nature Communications DOI: 10.1038/NCOMMS14396





