ABOUT
Our lab’s focus is on understanding the mechanisms that control mammalian embryo implantation and apply that knowledge to provide solutions that improve human assisted reproduction techniques (ART).
The embryonic development of humans (and mammals in general) requires the implantation of the embryo into the walls of the maternal uterus. This process is highly inefficient as on average, 25–30% of conceptions lead to successful live births and around 60% of all conceptions are lost at the time of (or soon after) implantation. However, despite the central role of implantation in human fertility, the process is still elusive to experimentation because of its inaccessibility.
To overcome the elusiveness of this process, the group combines imaging and bioengineering methods to efficiently culture and image pre-implantation embryos and allow them to implant outside the uterus in highly physiological conditions. Our systems are accessible to imaging tools which allow us to interrogate the genetics, metabolomics, and mechanics of the embryo in a high throughput manner. Using our systems, we are capable to (i) improve embryo culture conditions and (ii) diagnose embryos with improved implantation potential.
Label-free microscopy and multi/hyperspectral imaging
Label-free Microscopy: This technique allows for analyzing cells in their native condition, i.e. without being labeled or altered in any way, by means of retrieving cells autofluorescence signals and thus providing essential metabolic information about living tissues. Combined with multispectral methods, we delve deep into the metabolic complexity of embryos and oocytes, revealing insights previously unattainable.
Hyperspectral Imaging: Our groundbreaking approach employs hyperspectral imaging to obtain the metabolic profiles of embryos and oocytes. This methodology allows us to identify key characteristics at the metabolic level, invisible to conventional techniques like brightfield imaging, offering a unique window into fundamental biological processes.
Hardware Techniques: We have extensive expertise in a variety of advanced microscopy techniques, including two-photon microscopy, laser scanning confocal microscopy, spinning disk confocal microscopy and light-sheet microscopy. The integration of these techniques with multi/hyperspectral detection methods enables us to observe biological samples of interest at cellular level with spectral characteristics.
AI and Software Analysis: We employ sophisticated data analysis tools such as spectral histogram analysis and phasor-plot analysis combined with artificial intelligence (AI) methods for classification. These techniques allow us to interpret complex multi/hyperspectral data and draw meaningful conclusions about the viability and quality of embryos and oocytes.
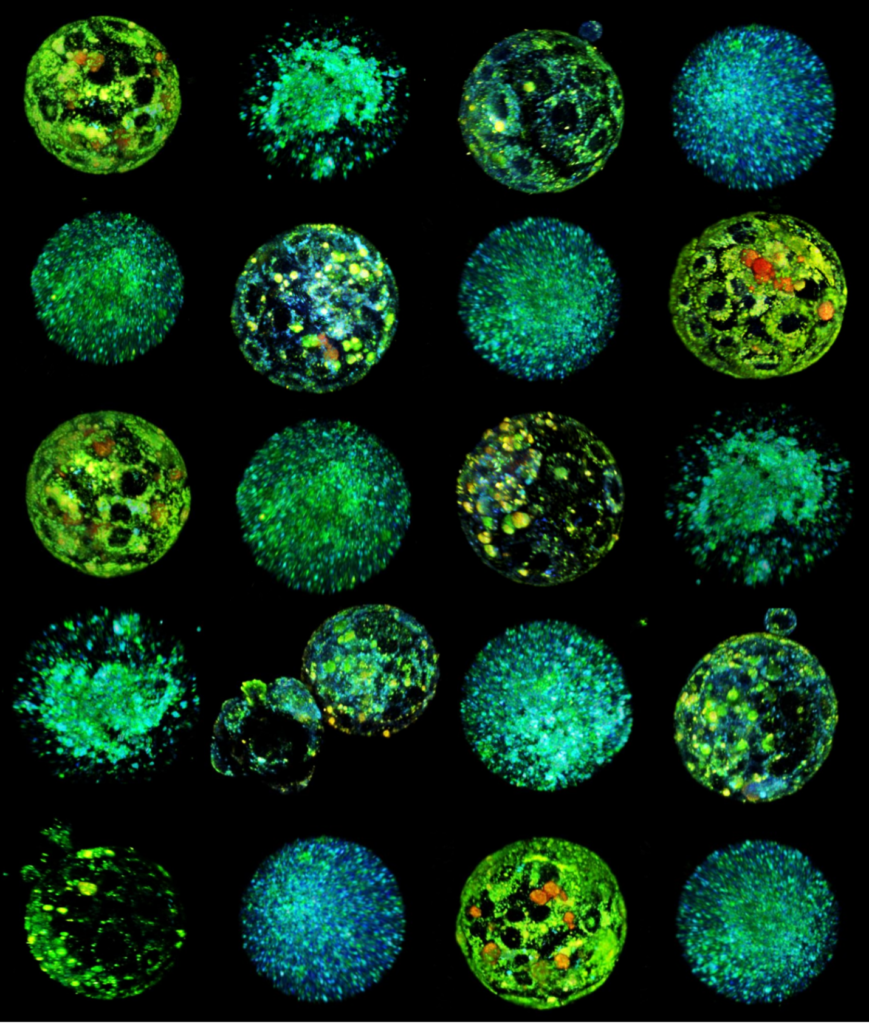
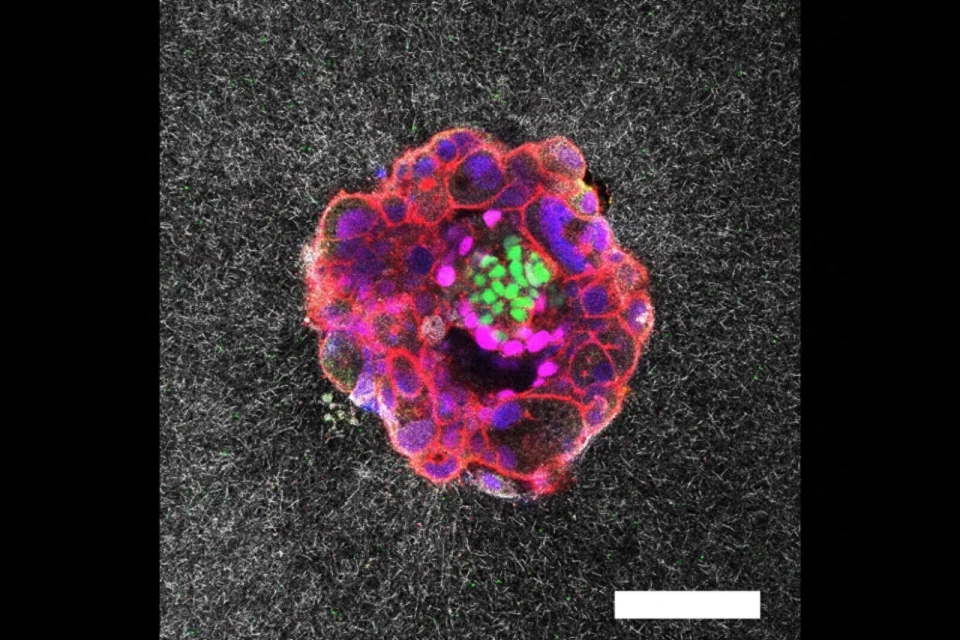
4D (x, y, z, and λ) Hyperspectral Autofluorescent Images of Mouse Oocytes and Blastocysts. The samples were imaged using a Zeiss LSM780 inverted microscope with a C-Apochromat 40x/1.20 W Korr Zeiss objective. The imaging conditions were maintained at 37°C and 5% CO2. A Mai-Tai DeepSee laser provided two-photon excitation at a wavelength λex = 780nm. The emitted autofluorescence spanned from 410 nm to 695 nm and was collected by a 32-channel PMT GaAsP spectral detector. The image captures multiple planes, separated by 2.5μm and 5μm steps for oocytes and embryos respectivelly. The pseudo-color representation of the hyperspectral images was achieved using additive blending to integrate the 32 channel colors for each pixel.
Bioengineering to improve embryo implantation
We have developed proprietary 3D ex vivo hydrogel-based implantation platforms which mimic the uterine microenvironment, allowing the embryo to progress towards post-implantation stages in an amenable way for optical microscopy. Working towards obtaining dynamic control of embryo culture, we have integrated our hydrogels in a microfluidic device allowing for controlled nutrient supply, oxygen concentration and long-term embryo culture. Our 3D ex vivo hydrogel-based implantation platforms allow for drug screening and determination of its impact on embryo implantation and development.
We use our 3D ex vivo implantation platform to understand embryo implantation with a focus on the biomechanics of the system. To this means we quantify the displacement of the matrix generated by the embryos using PIV or DVC algorithms. We look at the forces and resulting patterns embryos are applying in order to penetrate the hydrogel and also how external forces affect embryo implantation.
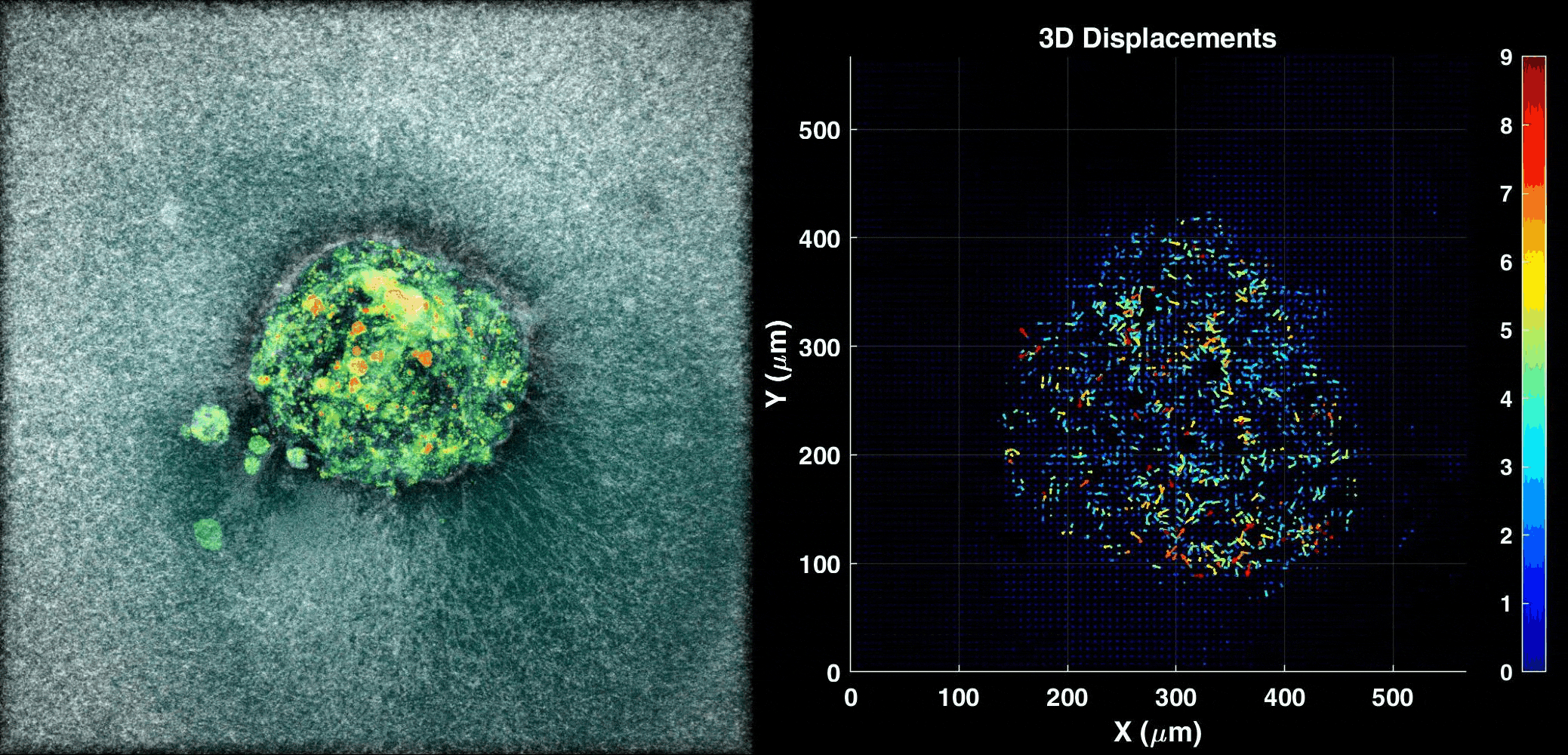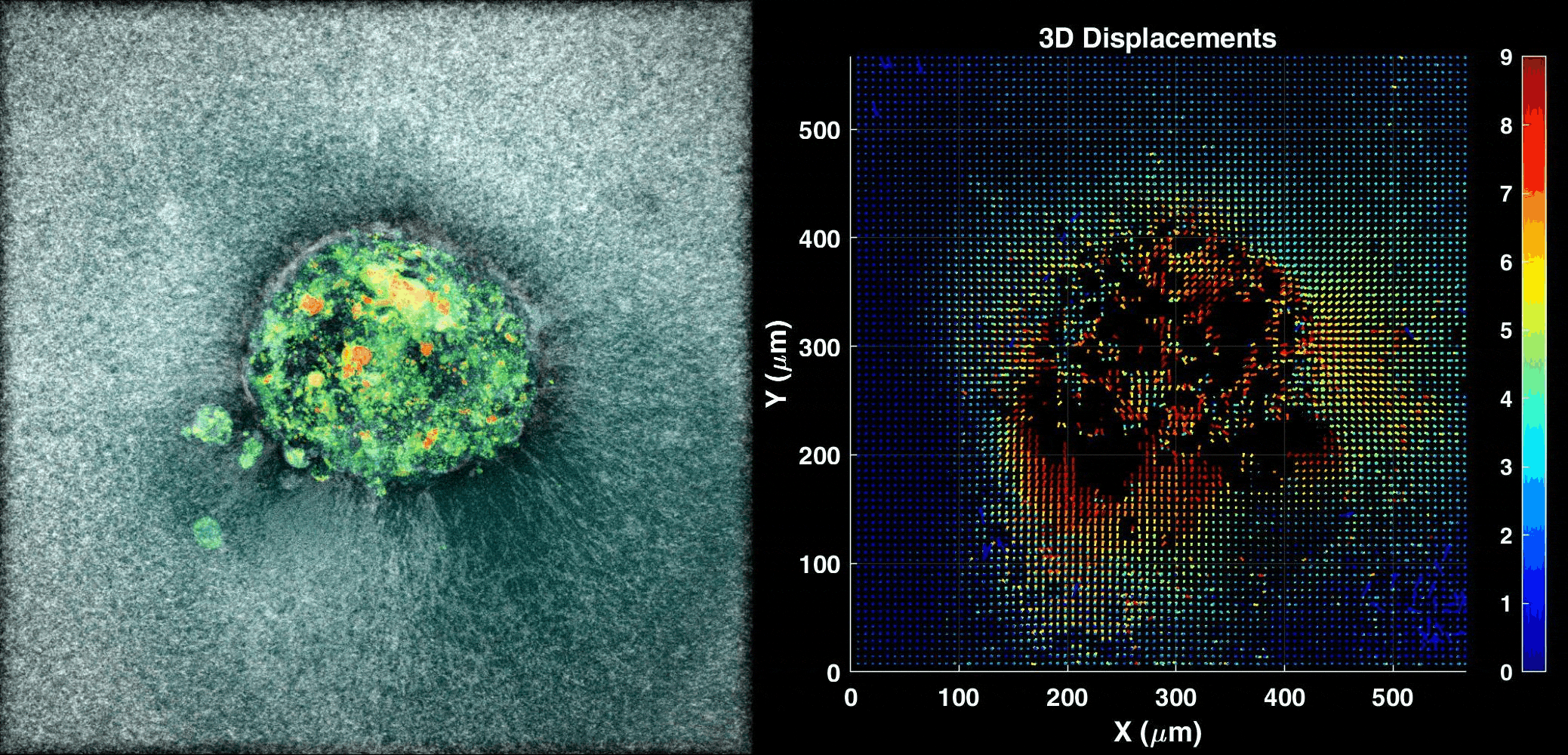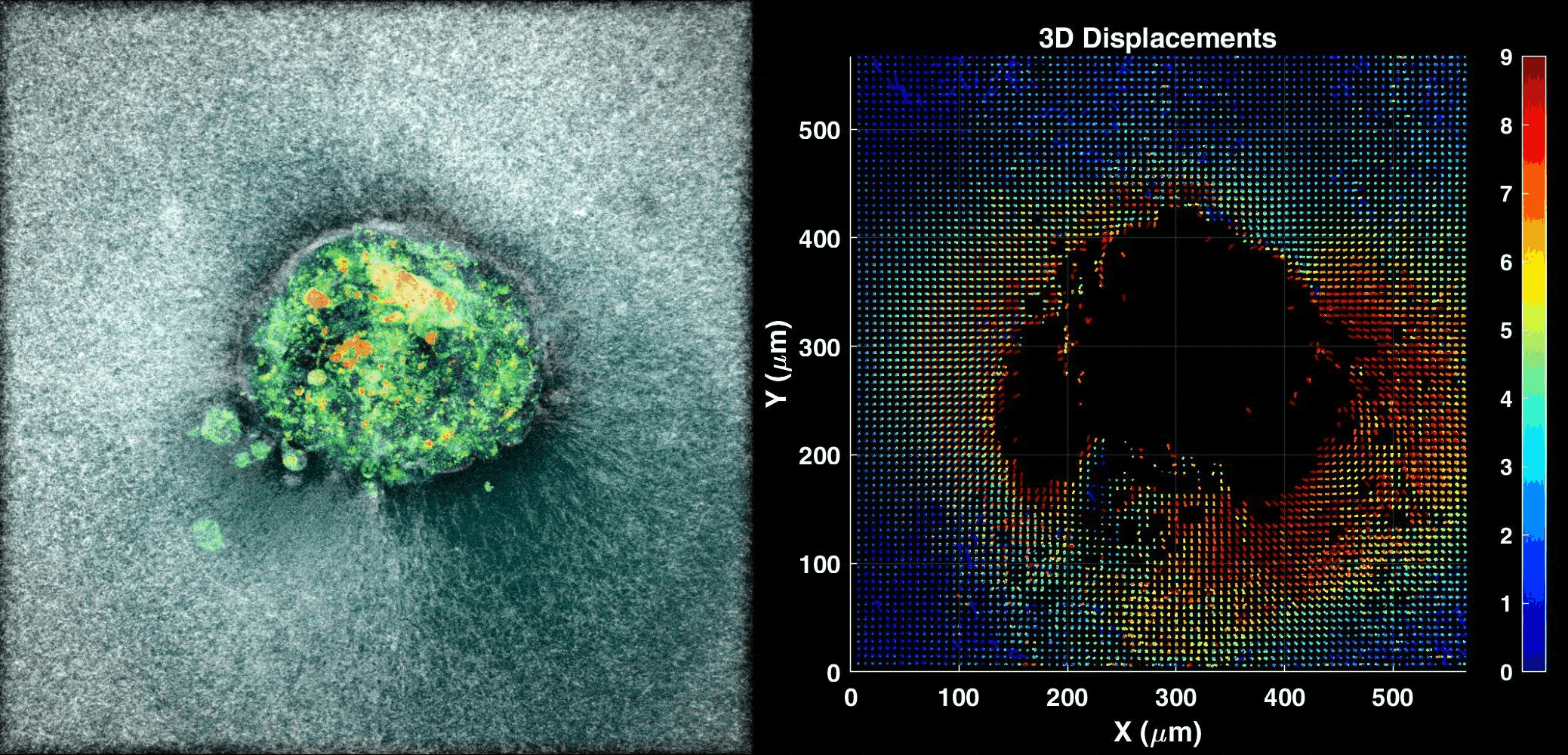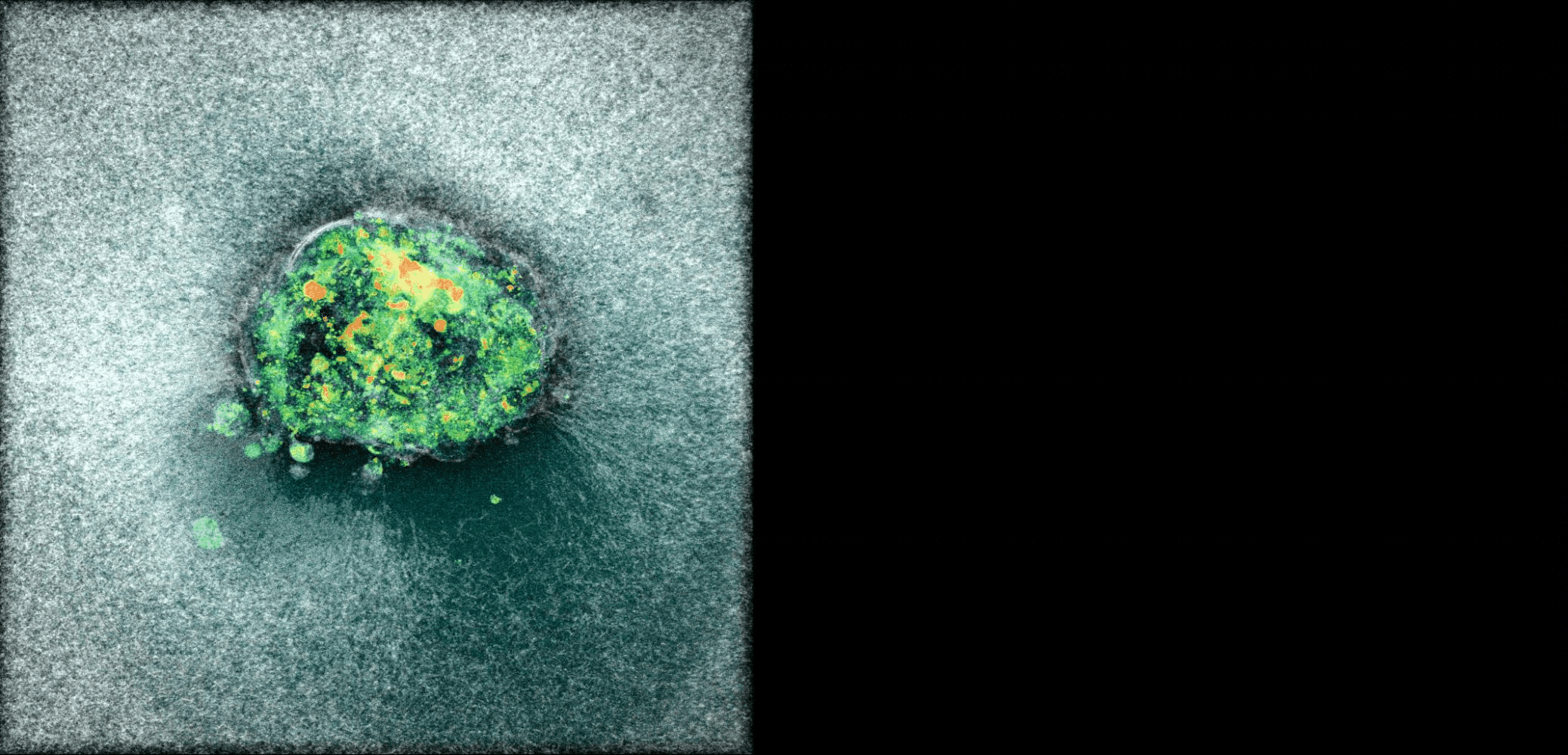
Human Embryo implanting on a 3D platform
Embryo Culture Supplements Development from Human Plasma
Traditionally, embryo culture relied on Human Serum Albumin (HSA) as a key protein component. However, HSA underrepresents the rich composition of proteins present in human plasma. We work with a new-generation of supplements, which go beyond mere albumin, encompassing essential components such as growth factors and globulins, crucial for fostering optimal embryo development. In our group, we test new supplements to enhance embryo development and implantation. Our clinical grade human-derived supplements improve blastulation and implantation rates both in human and mouse embryos, showing superior lineage segregation and spatial organization compared to control counterparts.
The Bioengineering in Reproductive Health is the first Open Innovation Lab research unit at IBEC.
Due to the high translational component of our research, we have established collaboration contracts with the pharma industry, hospitals, and venture capital to bring our technology to the clinics and the market. Our Open Lab is a multidisciplinary environment where embryologists, cell biologists, optical physicists, biophysicists, and business developers synergize to create a unique environment shaped by science and entrepreneurship.
STAFF
PROJECTS
European Projects
| HSMe-ImPredict · Development of non-invasive imaging methodology for improving embryo implantation prediction, via hyper-spectral metabolic profiling (2022-2024) | Marie Curie Individual Fellowship | Samuel Ojosnegros |
National projects
| HYSPLANT · Selección de embriones para fecundación in vitro: predicción del éxito de implantación mediante clasificación metabólica de embriones (2020-2023) | MINECO Retos investigación: Proyectos I+D | Samuel Ojosnegros |
| Prediction of implantation success by hyperspectral metabolic profiling of human embryos obtained by in vitro fertilization (2020-2022) | AGAUR Beatriu de Pinós 2018 | Samuel Ojosnegros |
Private sector
| Estudio del mecanismo de acción de OXO-001 (2020-2022) | Oxolife | Samuel Ojosnegros |
| Evaluación de derivados plasmáticos humanos para el cultivo de embriones (2019-2021) | IVFTECH S.L. | Samuel Ojosnegros |
| Evaluación de derivados plasmáticos humanos para el cultivo de células mesenquimales y CAR-T (2020-2022) | IVFTECH S.L. | Samuel Ojosnegros |
| Prediction of implantation success by single-cell transcriptomic profiling of human embryos obtained by in vitro fertilization (2018-2022) | Scranton Enterprises BV | Samuel Ojosnegros |
Finished projects
| HYSPLANT · Metabolic profiling of in vitro fertilization embryos using hyspectral imaging (2019-2020) | European Commission ATTRACT | Samuel Ojosnegros |
| Embryo on a chip: Smart microdevice development for high-throughput screening embryo implantation (2020) | Tecniospring 2020 | Maria Demestre Viladevall |
NEWS

IBEC showcases cutting-edge biomedical innovation at the Health Revolution Congress 2026
The Institute for Bioengineering of Catalonia brought together researchers, investors and industry leaders with its event “IBEC Health Revolution Day: Engineering the Future of Medicine”, as part of the Health Revolution Congress. Through expert panels, technology pitches and scientific talks, the event showcased cutting-edge biomedical technologies and discussed how to translate scientific discoveries into real-world healthcare solutions.

IBEC showcases its leadership in biomedical innovation at MWC26 with three disruptive projects
Last week, Barcelona played host to MWC26, the world’s premier technology and connectivity event. The Institute for Bioengineering of Catalonia was in attendance, showcasing some of its spin-offs and state-of-the-art biomedical technologies, including: Nanobots Therapeutics, Lumiris Spectral Solutions, and Drug4Sight.

BIST calls for reforms to turn scientific discoveries into European industry
The BIST Forum 2026 brought together scientists and representatives from academic, economic and social institutions to discuss how frontier research can help build Europe’s industries of the future. The event also announced the recipients of the BIST To the Mothers of Science and Ignite programmes.

IBEC secures competitive funding to drive forward validation testing for three innovative health projects
IBEC has been awarded three grants in response to the call for proposals from the Strategic Plan for Health Research and Innovation (PERIS). These projects aim to validate hyperspectral technology for improving assisted reproduction, using bioprinted cell spheroids to combat type 1 diabetes and developing light-activated drugs to restore vision in patients with retinal degeneration.

Bioengineering for precision medicine at the 18th IBEC Symposium
The 18th annual IBEC Symposium focused on ‘Bioengineering for Precision Medicine’, which is one of IBEC’s key areas of application. The event was attended by nearly 300 people, including local and international researchers. The multidisciplinary environment provided experts from other centres and the IBEC community with the opportunity to present their projects and exchange knowledge.

IBEC and EMBL Barcelona co-organise a matchmaking workshop to explore synergies
Today, the Institute for Bioengineering of Catalonia (IBEC) and the European Molecular Biology Laboratory (EMBL) held a matchmaking workshop. The event brought together leading researchers from both centres to encourage the formation of new connections and promote scientific dialogue.

El Periódico: El asalto del embrión al útero, al descubierto
Un vídeo hecho en Barcelona documenta por primera vez en un modelo artificial de matriz la implantación, momento crítico y gran desconocido del embarazo humano.
Corriere della Sera: Filmato per la prima volta l’impianto d’embrione umano, il «miracolo della vita» si mostra in tempo reale
Il momento clou della gestazione è stato catturato per la prima volta da alcuni ricercatori dell’Istituto di Bioingegneria della Catalogna. La nuova tecnica potrebbe aiutare gli scienziati a scoprire come prevenire gli aborti spontanei
Metrópoles: Vídeo: registro mostra implantação de embrião humano pela primeira vez
Imagens inéditas revelam como embriões humanos invadem o tecido uterino e explica causa comum de infertilidade
JOBS
Researcher in Training at the Bioengineering in Reproductive Health Research Group
Ref: RT-SO // Deadline: 24/11/2025
Scientific Liaison at the Bioengineering in Reproductive Health Research Group
Ref: SL-SO // Deadline: 16/06/2025
Postdoctoral Researcher at the Bioengineering in Reproductive Health Research Group/ Unit
Ref: PR_SO // Deadline: 13/11/2024
Embryology Laboratory Technician at the Bioengineering for Reproductive Health Research Group
Ref: PD-SO/Deadline: 04/04/2024
Open postdoc position on embryology at the Bioengineering in Reproductive Health Research Group
Ref: PD_SO/ Deadline: 12/02/2024
Research Assistant at the Bioengineering for Reproductive Health Research Group
Ref: RA_SO/Deadline: 20/11/2023
Postdoc on microscopy at the Bioengineering in Reproductive Health Research Group
Ref: PD_SO//Deadline: 1/11/2023
Postdoctoral researcher at the Bioengineering in Reproductive Health Open Lab (Ref: PD_SO)
Ref: PD_SO // Deadline: 19/01/2023
Embryologist at the Bioengineering in Reproductive Health Research Group (Ref: LT_SO)
Ref: LT_SO // Deadline: 19/01/2023
Biochemist (with expereince in hematology) at the Bioengineering in Reproductive Health Research Group
Reference: LT-SO / Until November 19th, 2022
PUBLICATIONS
Check for more detailed information on the outputs of the Group at IBEC CRIS portal.
Publications list:
EQUIPMENT
- Embryo culture laboratory
- IFV workstations in laminar flow hoods
- Microscope
- Micromanipulation-microinjection station
- Embryo biopsy laser
- K-systems incubator
- Cell culture laboratory
- Biosafety cabinets
- Incubators
- Automated cell counter
- Dry warming/thawing system
- Sterile tubing welder
- Tubing sealer
- Centrifuges
- Advanced live imaging: photoconversion, 3D imaging, light scattering, spectroscopy
- Crest spinning disk mounted on a Nikon Ti
- Image analysis workstation
COLLABORATIONS
- Prof. Anna Veiga – Barcelona Stem Cell Bank (IDIBELL) and Dexeus Mujer, Barcelona
- Dr. Montserrat Boada/ Dr. Pere Barri – Dexeus Mujer, Barcelona
- Dr. Ayelet Lesman – Tel Aviv University (TAU), Israel
- Dr. Elena Martínez – IBEC
- Dr. Francesco Cutrale, University of Southern California (USC), USA
- Dr. Manuel Irimia – CRG, Barcelona
- Dr. Javier Ramón – IBEC
ENTREPRENEURSHIP
· Jorge Fuentes,
Business Strategy, A_Ventures, Barcelona, Spain
 |  |  | |
 |  |
Follow us on Twitter: @Biorephealth