About
Currently, a myriad of drug carriers or delivery systems have been developed to improve the overall performance and safety of therapeutic agents. However, our ability to treat neurological maladies, genetic syndromes, cancer, etc., remains a major challenge. One of the prime obstacles is our limited knowledge on the biological parameters that regulate the interaction of these systems with our tissues and, hence, our inability to gain non-invasive, efficient, and specific access within the body, its cells, and subcellular organelles.
To generate knowledge and tools to overcome this obstacle, we study the biological mechanisms ruling how our cells and tissues transport cargoes to precise destinations within our bodies and apply this knowledge to the design of nanodevices for improved delivery of therapeutic agents to specific disease sites.
Biologically-Controlled Transport of Drug Carriers
Most current strategies aim to bind a drug nanocarrier to a specific cell-surface receptor; but after said binding takes place, receptor-associated signaling and transport processes take control. Instead, our laboratory can impart a drug carrier control over the biological events that occur beyond binding. We have shown how using the same receptor, the kinetics, mechanism, and destination of a drug carrier can be modulated by: (a) varying its size, shape, and targeting valency; (b) varying the receptor epitope to which the carrier binds; (c) coupling carriers to signaling molecules; (d) combining targeting to several receptors; or (e) coupling targeting moieties with anti-phagocytic moieties on the surface of drug carriers.
Transport of Drug Carriers Across Physiological Barriers
Crossing the linings that separate body and cellular compartments is paramount for efficient drug delivery. We were first to identify a natural pathway regulated by ICAM-1, a cell adhesion molecule overexpressed by most cells under pathological states, which enables transcytosis across the epithelial barrier that separates the gastrointestinal tract from the bloodstream for oral delivery, and the blood-brain barrier that separates the bloodstream from the brain tissue for delivery of therapeutics against neurological diseases. We also work with DNA-built nanocarriers which enable uptake within cells and endosomal escape for delivery to the cytosol and other subcellular compartments.
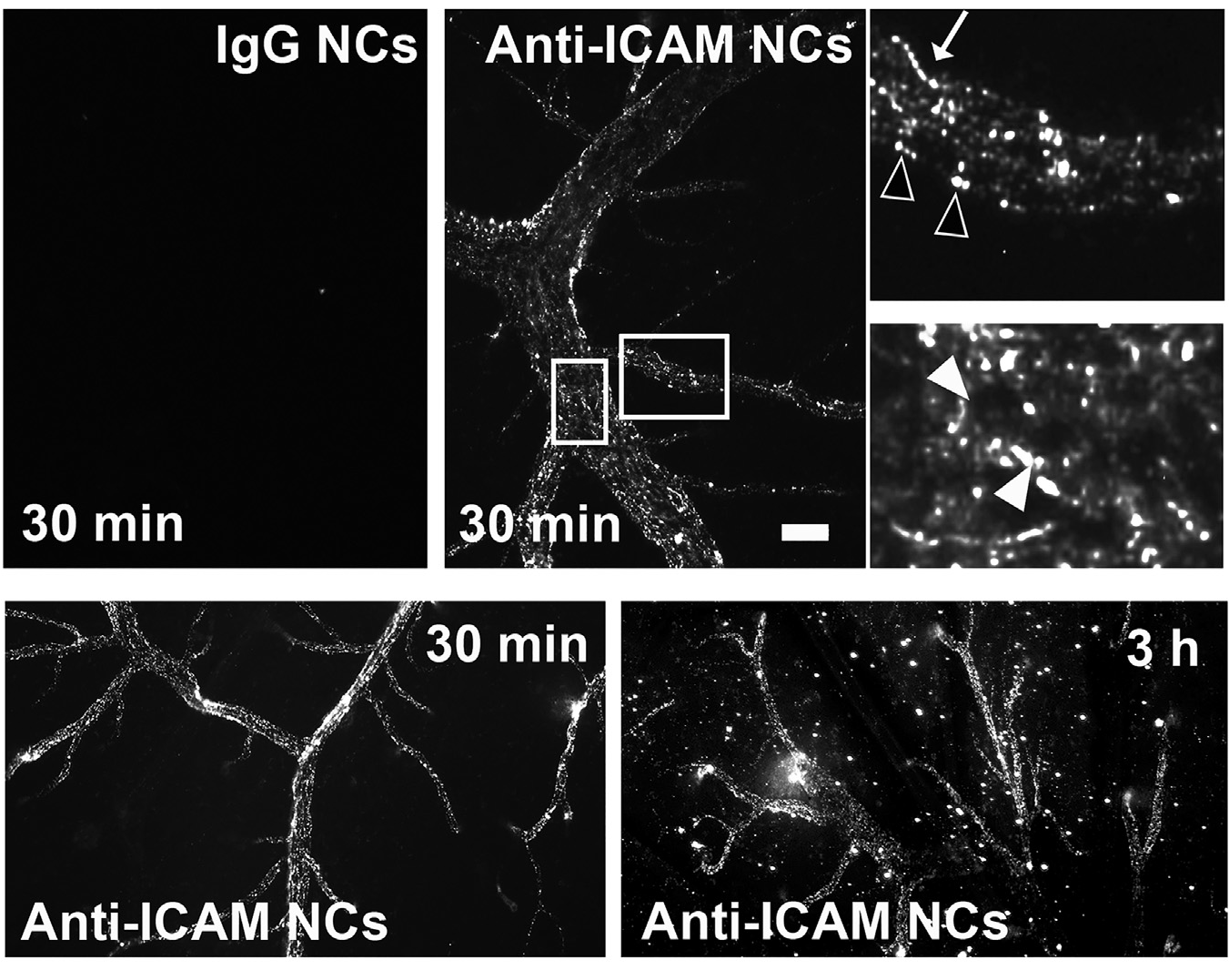
Figure 1. ICAM-1-targeted nanocarriers (anti-ICAM NCs) crossing the blood-brain barrier in an animal model. Fluorescence microscopy of cortical brain samples isolated from mice 30 min or 3 h after intravenous injection with green-fluorescent anti-ICAM NCs or control IgG NCs. Scale bar = 10 μm. Boxes = regions magnified 3-fold in the right panels. Arrows = NCs on the blood-facing surface of the blood-brain barrier. Open arrowheads = NCs on the brain-facing surface of the blood-brain barrier. Closed arrowheads = NCs inside of brain endothelial cells. Extracted from Manthe et al., Intertwined mechanisms define transport of anti-ICAM nanocarriers across the endothelium and brain delivery of a therapeutic enzyme. J Control Rel. 324 (2020):181-193).
Improving Treatment of Lysosomal Disorders
Pathologies due to monogenic deficiencies, such as the case of lysosomal disorders, are valuable models to study disease progression and therapeutic intervention because of their well-known etiology, unequivocal diagnosis, and availability regarding patient samples, diverse cell types, and animal models. Since these diseases associate to either acute or long-term effects depending on genetic severity, and they associate with neurodegeneration, cardiovascular, metabolic, and cancer-like syndromes, they represent excellent disease models. Consequently, we are applying targeted nanotechnology concepts to the treatment of genetic lysosomal disorders. Current therapies by i.v. enzyme infusion are only helpful for diseases where clearance cells and organs (liver, spleen, macrophages, etc.) are the main targets. Yet, delivery to other organs such as the brain, lungs, etc. hinders treatment for most of these diseases. Using types A and B Niemann-Pick, Fabry, and Gaucher diseases as examples, we are developing new therapeutic strategies to deliver therapeutic enzymes to all affected organs in animal models, which holds considerable translational potential.
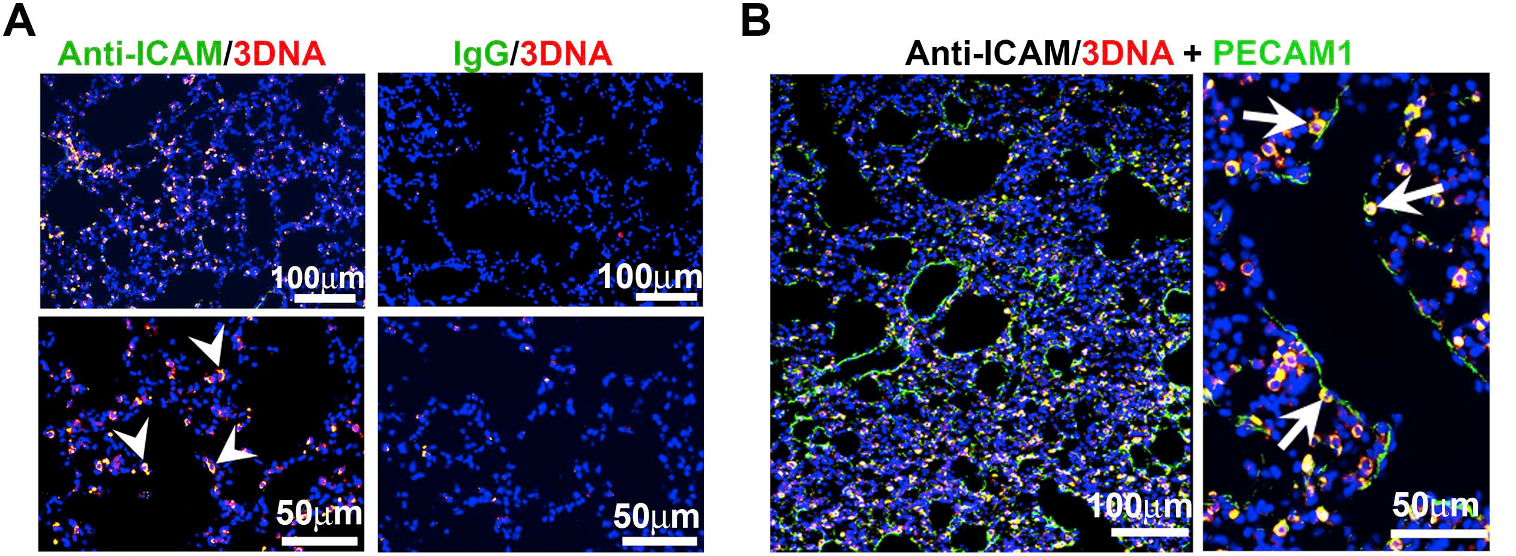
Figure 2. ICAM-1 targeted DNA-made nanocarriers (anti-ICAM/3DNA) for drug delivery to the lungs. Cy3-labeled 3DNA targeted to ICAM-1 (anti-ICAM/Cy3-3DNA) or non-targeted controls (IgG/Cy3-3DNA) were intravenously injected in mice. Lungs were isolated and processed at sacrifice at 60 min. (A) Confocal microscopy shows Cy3 3DNA (red), while the targeting coat (antibody) is visualized using a FITC-secondary antibody (green). Arrowheads show colocalization of the antibody on the coat of 3DNA. (B) Confocal microscopy showing anti-ICAM/Cy3-3DNA (red) and lung endothelial cells visualized using polyclonal anti-PECAM-1+FITC-secondary antibody (green). Arrows indicate colocalization of Cy3-3DNA with endothelial PECAM-1. (Extracted from Roki et al, Unprecedently high targeting specific to lung ICAM-1 using 3DNA nanocarriers. J Control Release. 305 (2019):41-49).
Staff
Silvia Muro
Projects
| NATIONAL PROJECTS | FINANCER | PI |
|---|---|---|
| CROSSTARGET · Desarrollo de nuevas herramientas traslacionales multi-especie para el direccionamiento de terapias con precisión de órgano y subcelular (2019 – 2022) | Ministerio de Ciencia, Innovación y Universidades | Silvia Muro |
| NANO-GBA · Assessing the effects of glucocerebrosidase (GBA) alterations on receptor membrane nanoarchitecture to design improved nanomedicines(2020 – 2021) | BIST Ignite Program | Silvia Muro |
| FUNDRAISING PROJECTS | FINANCER | PI |
|---|---|---|
| Campaña FasterFuture “A por el Parkinson” (2019 – 2021) | Fundraising | Silvia Muro |
| FINISHED PROJECTS | FINANCER | PI |
|---|---|---|
| BBB2GATE · Control diferencial del transporte de vehiculos terapeuticos dentro versus a traves de la barrera hematoencefalica (2018 – 2020) | MINECO | Silvia Muro |
Publications
Check for more detailed information on the outputs of the Group at IBEC CRIS portal.
Publications list:
Equipment
- Dynamic Light Scattering & ζ-Potential: Malvern’s Zetasizer Ultra.
Determination of the size, concentration, mobility, ζ-potential of nanoparticles, including high resolution size and MW distribution, non-invasive retrodispersion optics for wide concentration ranges, a fluorescent filter wheel to minimize artifacts from fluorescent samples, multiangle scattering (back, side, and forward scatter) to determine particle concentration without calibration, and constant current mode to measure high conductivity simples. - Gel Permeation Chromatography: Malvern’s OMNISEC.
Detector system (refraction index) of 640 nm y 50 mW, as well low (<10°) angle for highly sensitive determination of the absolute MW of polymers and proteins, upgradable to measure viscosity, branching, etc. Chromatography module, regulable pump, degasifier, sampler adaptable to many sample formats, temperature control for high MW polymers, columns/pre-columns for separation of polymers and proteins, and fraction collector. - Confocal-epifluorescence microscope: Zeiss LSM 700 microscope with 100 mW max output, 400-650 wavelength range, and IIIb class laser; 10x: EC Plan-NEOFLUAR, 20x: LD Plan-NEOFLUAR, 50x: EC Epiplan-NEOFLUAR, and 100x/1.3 oil: Plan-NEOFLUAR objectives; AxioCam Mrm camera; XLmulti S1 incubator system, and Zen 2 blue software.
Collaborations
- Dr. Alexander Andrianov, University of Maryland, MD, USA.
- Dr. Yu Chen, University of Maryland College Park, MD, USA.
- Dr. Mandy Esch, National Institutes for Standards and Technology, Gaithersburg, MD, USA.
- Dr. Robert Getts, Genisphere LLC, Hatfield, PA, USA.
- Dr. Hamid Ghandehari, University of Utah, UT, USA.
- Dr. Joe Kao, University of Maryland Baltimore, MD, USA.
- Dr. Peter Kofinas, University of Maryland College Park, MD, USA.
- Dr. Juan Marugan and Dr. Wei Zheng, National Institutes of Health, Rockville, MD, USA.
- Dr. Vladimir Muzykantov, University of Pennsylvania, Philadelphia, PA, USA.
- Dr. Gianfranco Pasut, University of Padova, Padova, Italy.
- Dr. Edward Schuchman, Mount Sinai School of Medicine, New York, NY, USA.
- Dr. Brigitte Stadler, Aarhus University, Denmark.
- Dr. Alberto Fernández de las Nieves, ICREA & University of Barcelona, Spain.
- Dr. Dolores Ledesma, CBM & CSIC, Madrid, Spain.
- Dr. Ricardo Feldman, University of Maryland, Baltimore, MD, USA.
- Dr. Ramón Farré, Clinic Hospital & University of Barcelona, Spain
- Dr. Jesús Villarrubia, Ramón y Cajal Hospital, Madrid, Spain
- Dr. Josep Samitier, IBEC & University of Barcelona, Spain.
- Dr. Giuseppe Battagglia, ICREA & IBEC, Barcelona, Spain.
- Drs. Marina Giannotti & Pau Gorostiza, IBEC & ICREA, Barcelona, Spain.
- Dr. Juana Díez, Pompeu Fabra University, Barcelona, Spain.
News
Identified the best routes to bring drugs inside the brain during the neurological pathology Niemann-Pick disease type A
Researchers identified how a neurological disease from the group of the lysosomal storage disorders, alters the natural entrance routes of molecules in the brain. They identified the most active ways … Read more

The Catalan Association for Parkinson’s visits IBEC
On November 18th, members of the Catalan Association for Parkinson’s visited the Institute for Bioengineering of Catalonia (IBEC) to see, first-hand, the work of Silvia Muro and her research group, which use nanotechnology to find solutions to this disease. Professor Muro’s work has been supported, among others, by donations received through the IBEC “Faster Future” program.

Faster Future campaign against Parkinson’s culminates with new scientific breakthroughs
IBEC Faster Future program has made possible, over the last two years, and thanks to donor contributions, the study of antibodies as the basis of a therapeutic product for the treatment of Parkinson’s. The researchers now present the scientific advances and will continue working to try to respond to one of the greatest challenges of modern medicine: neurodegenerative pathologies such as Parkinson’s.

IBEC Group Leader Silvia Muro, elected to join the American medical and biological engineering elite
The American Institute for Medical and Biological Engineering (AIMBE) has announced the election of Dr. Silvia Muro as a member of the college of Fellows for her achievements on the behavior of nanomedicines at the cellular and molecular levels.

IBEC researchers find a new way to effectively transport drugs to the brain
An international group of researchers from the University of Maryland (United States) and the Institute for Bioengineering of Catalonia (IBEC) led by ICREA Research Professor Silvia Muro, has identified a new way of transporting drugs to the brain, one of the major challenges of the pharmaceutical science today, that could help to come up with new treatments for neurological diseases such as Parkinson’s or Alzheimer’s. To find this out, the experts linked an antibody capable of recognizing the ICAM-1 protein -a molecule expressed on the surface of blood vessels- to a series of polymeric nanoparticles that can transport drugs and inject them intravenously.

The second edition of IBEC’s Faster Future programme will be dedicated to the fight against Parkinson
The programme IBEC Faster Future, an initiative that aims to help accelerate research projects that are close to tackling major challenges in health, will enable the development of a new antibody that will be the base of a therapeutic product for the treatment of Parkinson’s disease. The Faster Future program campaign “Let’s tackle Parkinson”, which is launched today and will remain open to donations until the 30th of April, has the aim to raise 50,000 € needed to accelerate this research, seeking to obtain favourable results within a year and a half.

IBEC project to defy blood-brain barrier awarded in highly competitive call
IBEC group leader Silvia Muro has been granted funding in MINECO’s ‘Explora Ciencia’ and ‘Explora Tecnología’ 2017 call. It’s the first competitive grant for Silvia and her group since she joined IBEC at the end of 2017, and one of only 97 research projects to be financed out of the 1594 applications submitted – a success rate of only 6%. The project, ‘Controlling the differential transport of therapeutic cargoes into versus across the BBB (BBB2GATE)’ will aim to develop drug vehicles that can cross the blood-brain barrier using the natural routes that the body’s substances use to circumvent this obstruction.
Jobs
Predoctoral researcher at the Targeted Therapeutics & Nanodevices Research Group (Project BRAINZYME)
Ref: FPI_SM // Deadline: 29/10/2025
Research Assistant at the Targeted Therapeutics & Nanodevices Research Group
Application Deadline: 02/12/2021Ref: RA-SM The Targeted Therapeutics and nanodevices group at the Institute for Bioengineering of Catalonia (IBEC) is looking for a Research Assistant to collaborate in a research project focused on drug delivery systems capable of targeting and crossing the blood-brain barrier (BBB) for treatment of neurological diseases involving lysosomal alterations and/or inflammation.


 ibecbarcelona.eu
ibecbarcelona.eu