About
Our research interests are focused on three main aspects of developmental neurobiology and neurodegeneration: development of new lab on chip devices for neurobiological research, new strategies to avoid α-synuclein and tau transport in neurons and new approaches to enhance axon regeneration after lesion.
Development of new lab on chip devices for neurobiological research
One of our focus is to mimic the developing and neurodegenerating nervous system in lab on chip devices. We believe that combining several different stimuli in the chip resembles a more realistic environment that nerve cells will encounter in the living animal in normal and disease conditions. These experiments using la bon chip models in turn will make future studies on the role of neuronal cells in development and regeneration more accurate and complete. Thus, we developed a new device able to reproduce the formation of the neuromuscular junction (NMJ) in lab on chip devices. In addition, a device designed to analyse axon lesions of cortical neurons was also developed.
We developed a new device able to reproduce the formation of the neuromuscular junction (NMJ) in lab on chip devices.
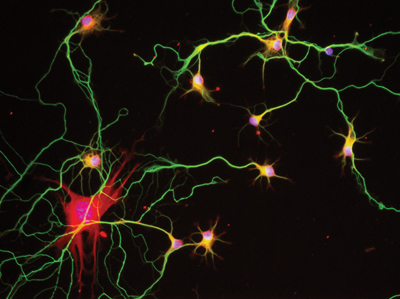
Current experiments of our group in collaboration with groups of IBEC, CIBER-BBN, and other labs aimed at developing new lab on chip devices to mimics and modulate particular neurobiological processes. For example: i) on chip lab platform to monitor drug delivery modulating neuronal activity (see figure); ii) cortico-spinal chips to develop axon regenerative studies of new drug formulation (in collaboration with Imperial College; London); iii) devices tmigrating neurons (in collaboration with i3A, Zaragoza) and, iv) in silico 3D modeling for neurodegenerative diseases (Alzheimer and Parkinson chip).
Last experiments are focused to reproduce a lab on chip devices to analyse and to reproduce ALS models (in collaboration(Alzheimer and Parkinson chip). Last experiments are focused to reproduce a lab on chip devices to analyse and to reproduce ALS models (in collaboration with Biodonostia Hospital) (Figure 2).
New strategies to avoid α-synuclein and tau transport in neurons
α-Synuclein is a key player in the pathogenesis of synucleinopathies, including Parkinson’s disease, dementia with Lewy bodies, and multiple system atrophy. Transmission of synthetic α-synuclein aggregates has been demonstrated in several cellular and animal models. Several groups have reported that insoluble α-synuclein shows “prion-like” propagation in wild-type mice. However, the basis of the spreading process remains poorly understood although cell to cell transport via exocytosis has been proposed. We described in 2018 that PrPC is a new receptor for α-synuclein involved in their spreading and propagation. Our new objectives aimed to block this interaction to reduce the neuropathological transport of α-synuclein. Similar experiments are also developed in the case of tau, one of the hallmarks of Alzheimer´s disease, since tau also binds to PrPC during its inter-neuronal propagation.
New approaches to enhance axon regeneration after lesion
Following axonal lesions in the adult CNS, damaged axons degenerate, while surviving fibres are unable to regenerate, and have a limited capacity to sprout and to re-establish lost connections. Experimental evidence suggests that providing a permissive extracellular environment containing cellular or biomaterial bridges is not enough to increase axon regrow to obtain functional regeneration, supporting the importance of clarifying the complex transcriptional response below these signalling pathways. As an alternative, activating or recapitulating the developmental program of the lesioned neuron to sprout and regenerate may represent a complementary therapeutic approach and may sometimes directly counteract the inhibitory signalling. Optogenetics is currently the state-of-the-art method for activity-based nervous system research, allowing more specific cellular stimulations by light in a less invasive techniques compared to classical electrical stimulation, but leading to a more tailored physiological response3. In our laboratory, we started to analyse whether optical stimulation of lesioned neurons is able to enhance axonal growth or sprouting. These experiments were performed on lesioned cortical neurons in vitro and in vivo.
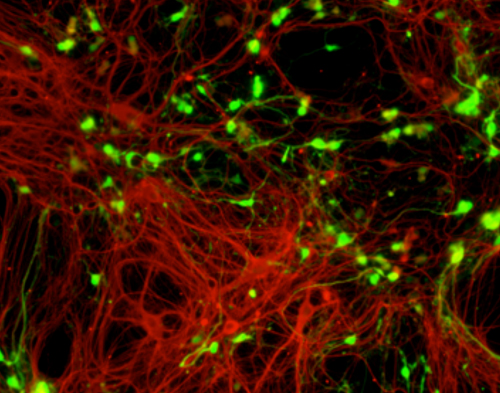
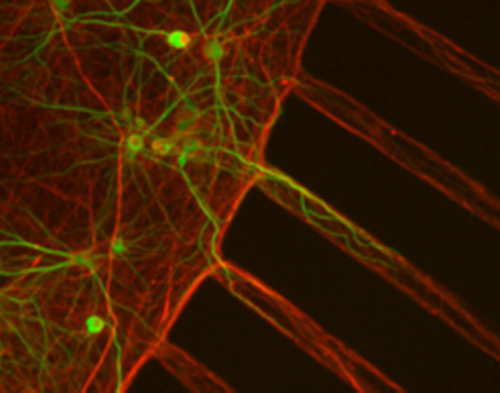
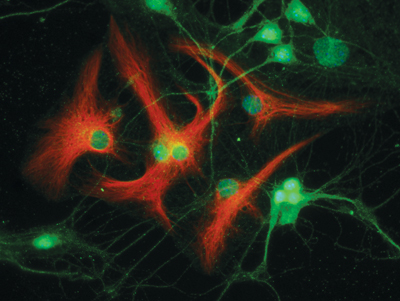


Staff
- Jordi Duran Castells, associated researcher
Projects
| NATIONAL PROJECTS | FINANCER | PI |
|---|---|---|
| PRPSEM · Nuevas aproximaciones para entender las funciones de la PrPC y miembros secretables de semaforinas durante el desarrollo del hipocampo y en neurotransmisión (2019-2022) | MICIU (Retos investigación: Proyectos I+D) | José Antonio Del Río |
| STOPTAUPATHOL · Modulation of Tau seeding and pathology in tauopathies by BBBnanocarriers, epitope selective vaccination and ectoPrP Tau receptor bodies (2019-2022) | Obra Social “La Caixa” | José Antonio Del Río |
| HDAC3i-SCI-EAE Inhibición de la HDAC3 como terapia para lesiones medulares y enfermedades autoimmunes demielinizantes (2021-2024) | MICIU (Retos investigación: Proyectos I+D) | Arnau Hervera |
| FINISHED PROJECTS | FINANCER | PI |
|---|---|---|
| DEVREG Nuevas funciones de PlexinD1/Sema3E, PrPC y las proteínas asociadas a la mielina durante el desarrollo de la corteza cerebral de roedores y en neurodegeneración (2013-2016) | MINECO Investigación fundamental no orientada | José Antonio Del Río |
| Red Nacional de Priones (2015-2017) | MINECO, Acciones Dinamización “Redes Excelencia” | José Antonio Del Río |
| NEURODEV Nuevas funciones de PlexinD1/Sema3E, PrPC y las proteínas asociadas a la mielina durante el desarrollo de la corteza cerebral (2013-2016) | MINECO, I+D-Investigación fundamental no orientada | José Antonio Del Río |
| Mecanismos epigenéticos implicados en la etiología y progresión de las demencias neurodegenerativas rápidamente progresivas (2015-2016) | MINECO | José Antonio Del Río/Miguel Calero |
| Monitoring neurocognitive deficits in Alzheimer’s and Parkinson’s diseases using saliva or blood-derived biomarkers and a multiplexed approach | Obra Social La Caixa | José Antonio Del Río |
| Spanish Network of Neurodegerative Diseases of the Ministry of Heath (FIS) (2009-2017) | CIBERNED, Reference: P1-L14 | José Antonio Del Río |
| ANGIODEVSNC Funciones de genes implicados en angiogénesis y remodelación vascular durante el desarrollo cortical y en neurodegeneración (2016-2019) | MINECO, Retos investigación: Proyectos I+D | José Antonio Del Río |
| Role of the cellular prion protein as “cross-talk” protein between a-syn/ LRRK2 and p-Tau in sporadic and familiar Parkinson’s disease (2015-2018) | Fundació La Marató de TV3 | José Antonio Del Río |
| Robots biológicos basados en el control de la union neuromuscular (2016-2019) | MINECO, Programa EXPLORA, Reference: TEC2015-72718-EXP | Josep Samitier |
| Red de Excelencia Nacional de Priones (2016-2018) | MINECO, Reference: AGL2015-71764-REDT | José Antonio Del Río |
Publications
Check for more detailed information on the outputs of the Group at IBEC CRIS portal.
Publications list:
Equipment
- Neural stem cell culture
- Microscopy facility (Olympus BX61 and Olympus IX71 with LCi culture and OKOlab systems)
- Electroporation system (BTX 600)
- Pressure microinjection system
- Protein expression and purification systems
- Technology of neuronal culture facilities (2D and 3D)
- Lentiviral production and characterization
- Protein and DNA electrophoresis
- In situ hybridization oven
- Optogenetic in vitro and in vivo stimulation system
Collaborations
- Dr. Adolfo Lopéz de Munain
Hospital de Donostia, San Sebastian, Spain - Dr. Joaquin Castilla
CiC Biogune, Bilbao, Spain - Prof. Juan María Torres
INIA-CISA CSIC, Valdeolmos, Madrid, Spain - Prof. José María Delgado and Prof. Agnest Gruart,
UPO, Sevilla, Spain - Prof. Jose Manuel García Verdugo
Facultad de Ciencias, Universidad de Valencia, Spain - Prof. Jose Manuel García Aznar
Nanotechnology Institute, Zaragoza, Spain - Prof. Fernando Albericio and Ernest Giralt
Institute for Research in Biomedicine (IRB), Barcelona - Dr. Miriam Royo
Institute for Research in Biomedicine (IRB), Barcelona - Dr. Elisabeth Engel, Prof. Josep Samitier, Prof. Xavier Trepat and Prof. Daniel Navajas
IBEC - Prof. Ángel Raya
Center of Regenerative Medicine in Barcelona (CMRB) - Dr. Antonella Consiglio and Dr. Franc LLorens
Institut d’Investigació Biomèdica de Bellvitge, University of Barcelona, Spain - Prof. Jesús Ávila and Prof. Francisco Wandosell
Consejo Superior de Investigaciones Científicas (CSIC), Universidad Autónoma de Madrid, Spain - Prof. Isidro Ferrer
Institut d’Investigació Biomèdica de Bellvitge, University of Barcelona, Spain - Dr. Alberto LLeó
Hospital Sant Pau, Barcelona, Spain - Prof. Miquel Vila
VHIR, Barcelona, Spain - Prof. Fanny Mann
Developmental Institute of Marseille Luminy, Université de la Méditerranée, Marseille, France - Prof. Yutaka Yoshida
Division of Developmental Biology, Cincinnati Children’s Research Foundation, Cincinnati, Ohio, USA - Prof. Masato Hagesawa
Faculty of Medicine, Tokyo - Prof. José Luis Lanciego
CIMA, Navarra, Spain
News

IBEC, one of the centres selected by the Pasqual Maragall Foundation for its research grants
Amaya Hernández-Vega, a senior researcher at IBEC, will lead one of the two projects selected in the 2024 call for proposals from the Pasqual Maragall Research Programme, which is dedicated to funding research into Alzheimer’s disease and other neurodegenerative conditions. IBEC also celebrates the participation of Jordi Duran, an associate researcher from the Molecular and Cellular Neurobiotechnology group, who will collaborate on the second selected project.

Bioengineering for Emergent and Advanced Therapies at the 17th IBEC Symposium
IBEC’s 17th Annual Symposium focused on ‘Bioengineering for Emergent and Advanced Therapies’, one of IBEC’s key application areas. Around 300 people attended the event, including local and international researchers. It was a multidisciplinary environment in which experts from other centres and the IBEC community itself had the opportunity to present their projects and share knowledge.

New biomarker to diagnose Alzheimer’s in asymptomatic stages
This is a molecule that is directly linked to the expression of the cellular prion protein found on the surface of nerve cells. The study, led by the IBEC, opens the door to the early detection of Alzheimer’s disease in asymptomatic stages in blood samples, favouring its diagnosis and treatment.

Laboratory Technician at the Molecular and cellular neurobiotechnology Research Group
Application Deadline: 22/07/2022Ref: LT_TR The Molecular and cellular neurobiotechnology group at the Institute for Bioengineering of Catalonia (IBEC) is looking for Laboratory Technician to contribute to the development of novel methods for Neurodegenerative studies using microfluidics and in vitro platforms. The contract will be within the framework of the project “Fabrication of novel microfluidic devices and applications to monitor tau-related neurodegeneration in vitro”.

Mechanical signals in the development of the cerebral cortex
Researchers at the Institute for Bioengineering of Catalonia (IBEC), coordinated by principal investigator José Antonio del Río, are in the media for a study in collaboration with the University of Barcelona (UB) that describes the role of mechanical signals in the formation of the cerebral cortex.

Mechanical signals involved in the early development of the mouse cerebral cortex described for the first time
A scientific team coordinated by José Antonio del Río from the Institute for Bioengineering of Catalonia (IBEC) observed the mechanical signals and traction forces involved in the migration of Cajal-Retzius cells, a group of neurons that play an important role in the development of the cerebral cortex. The study, conducted on rodents, has been published in the journal Frontiers in Cell and Developmental Biology.

The lack of cellular prion protein might lead to epilepsy symptoms and learning deficits
Researchers at IBEC, in collaboration with hospitals and international partners, have observed that the deficiency of the cellular prion protein leads to behavioral deficits, learning impairment, and enhanced epilepsy-like excitability of the brain, in mice.
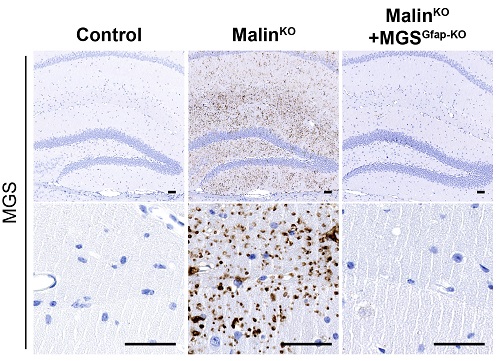
IBEC researchers contribute to discover the cause of epilepsy and neurodegeneration in Lafora disease
The accumulation of abnormal glycogen in glial cells of the nervous system causes inflammation and degeneration of the brain. The study, led by IRB Barcelona and with contribution of Arnau Hervera and Jose Antonio del Río from IBEC, has been recently published in the journal Brain.
Jobs
Laboratory Assistant at the Molecular and Cellular Neurobiotechnology Group
Ref: LA-TR // Deadline: 28/10/2025
Research in Training at the Molecular and Cellular Neurobiotechnology Group
Ref: RT-TR // Deadline: 27/10/2025
Predoctoral researcher at the Molecular and Cellular Neurobiotechnology Group (Project THRIVE)
Ref: FPI_JADR_RG // Deadline: 29/10/2025
Laboratory Technician at the Molecular and Cellular Neurobiotechnology Research Group (LT2-JADR)
Ref: LT2_JADR // Deadline: 13/11/2023
Laboratory Technician at the Molecular and Cellular Neurobiotechnology Research Group (LT-JADR)
Ref: LT_JADR // Deadline: 23/10/2023
Predoctoral researcher at the Molecular and Cellular Neurobiotechnology Group (Phd_JADR)
Ref: Phd_JADR // Deadline: 30/09/2023
Postdoc researcher at the Molecular and Cellular Neurobiotechnology Group (Ref: PD-TR)
Ref.: PD-TR / Deadline: 10/02/2023
PhD Researcher at the Molecular and Cellular Neurobiotechnology Group (Ref: PhD-TR)
Ref.: PhD-TR / Deadline: 30/01/2023
Laboratory Technician at the Molecular and Cellular Neurobiotechnology Research Group (Ref: LT-TR)
Ref: LT-TR // Deadline: December 26
Laboratory Technician at the Molecular and cellular neurobiotechnology Research Group
Application Deadline: 22/07/2022Ref: LT_TR The Molecular and cellular neurobiotechnology group at the Institute for Bioengineering of Catalonia (IBEC) is looking for Laboratory Technician to contribute to the development of novel methods for Neurodegenerative studies using microfluidics and in vitro platforms. The contract will be within the framework of the project “Fabrication of novel microfluidic devices and applications to monitor tau-related neurodegeneration in vitro”.


 ibecbarcelona.eu
ibecbarcelona.eu