About
The goal of our research is to gain a deeper understanding of cellular and respiratory biomechanics to improve the diagnosis and treatment of respiratory diseases.
The work is organized in two interrelated areas, focused on respiratory mechanics at both the systemic and the cellular level. We use basic and translational approaches in a multidisciplinary framework involving close cooperation with clinical groups.
Our current research interest is focused on the study of cell-matrix mechanical cross-talk for tissue engineering and regenerative medicine.
Cells sense and actively respond to the biophysical features of their microenvironment. Mechanical properties of the extracellular matrix regulate critical cell processes such as contraction, migration, proliferation, gene expression and differentiation.
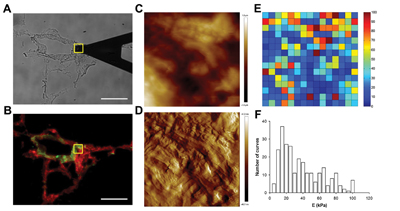
We use atomic force microscopy and other cutting-edge biophysical techniques to study the mechanical properties of the extracellular matrix and their impact in cell behavior. We have implemented protocols to decellularize different soft tissues. This innovative approach allowed us to reveal the local mechanical properties of the lung and heart extracellular matrix. By seeding cells in these scaffolds we study the impact of the mechanical features of the microenvironment on stem cell engraftment and differentiation onto lung and heart phenotypes. We produce lab-on-chip devices mimicking the native cell microenvironment to investigate mechanical signaling driving stem cell differentiation under precisely controlled conditions.
Using 3D bioprinters we integrate stem cells into synthetic and extracellular matrix hydrogels to fabricate tissue patches as an innovative approach to regenerate ventricular scars resulting from heart infarct. Organ biofabrication reengineered from decellularized tissue scaffolds offers a promising alternative for transplantation. We develop improved bioreactors mimicking breathing and blood perfusion to biofabricate lungs by seeding stem cells into acellular lung scaffolds.
Projects
| INTERNATIONAL PROJECTS | FINANCER | PI |
|---|---|---|
| Phys2BioMed · Biomechanics in health and disease: advanced physical tools for innovative early diagnosis (2019-2022) | European Commission, MARIE CURIE – ITN | Daniel Navajas |
| FINISHED PROJECTS | FINANCER | PI |
|---|---|---|
| MatriMec · Estudio multiescala de la mecànica no lineal de la matriz extracelular de pulmón (2018-2020) | MINECO, Proyectos I+D Excelencia | Daniel Navajas |
| Cardiopoesi amb biomatrius per regenerar la cicatriu post infart: From bench to bedside (first-in-man trial) | Pla Estratègic de Recerca i Innovació en Salut (PERIS) | Daniel Navajas |
| Señalización mécanica en la diferenciación de células madre en el pulmón. Modelo pulmón-en-un-chip | FIS-Proyectos de investigación en salud | Daniel Navajas |
| Bench test on performance of portable automatic CPAP devices (2016-2017) | RESMED (FBG2016A) | Ramon Farré (UB) |
| Precondicionamento biofísico de células madre mesenquimales para el tratamiento de la lesión pulmonar aguda provocada por sobreventilación en modelo animal 2015-2017) | FIS-MINECO (PI14/00280) | Daniel Navajas |
Publications
See full publication list in ResearcherID (h-index 51), Google Scholar (h-index 67) and ORCID
News
Daniel Navajas: 30 years dedicated to mechanobiology
Last May 5, on the occasion of Professor Daniel Navajas’ retirement, the IBEC held the symposium Before Mechanobiology had a name. The event paid tribute to the IBEC researcher’s exciting … Read more
A low-cost ventilator for areas with limited means
A project led by the University of Barcelona to which IBEC Group Leader Daniel Navajas has contributed has created a non-invasive low-cost ventilator to support patients with respiratory diseases in areas with limited means. Non-invasive ventilators are usually used to treat patients with respiratory failure: for example, those with severe complications due to COVID-19.
PeriCord, a bioimplant for infarcted hearts
The journal “EBioMedicine” of “The Lancet” has just published the procedure that allowed the creation, last year, of the “PeriCord”, the first human cardiac bioimplant, in which development the Institute for Bioengineering of Catalonia (IBEC) played a key role. In May 2019, a collaboration between the ‘Germans Trias i Pujol’ Hospital, the Blood and Tissue Bank (BST) and IBEC took a step forward for heart patients combining medicine, science and engineering.
First bioimplant with stem cells to regenerate the heart
The team led by Daniel Navajas, principal investigator at IBEC, together with the Blood and Tissue Bank have developed a bioimplant of stem cells of about 16 cm² that is applied directly to the heart. The bioimplant is composed of decellularised human pericardium and enriched with stem cells from umbilical cord, of great plasticity and, therefore, capable of regenerating tissues.
IBEC bioengineers contribute to a heart implant
A group from the Institute for Bioengineering of Catalonia (IBEC) led by Daniel Navajas contributes to an operation designed to repair the heart tissue of a 70-year-old patient who had suffered a heart attack. This was made possible by the creation of a bioimplant enriched with stem cells. The operation is the result of the joint work of scientists, doctors and engineers over more than ten years.
Sleep apnea could promote tumor growth in the young
A study published in the American Journal of Respiratory and Critical Care Medicine has revealed that sleep apnea could promote the growth of lung cancer in younger individuals. Researchers from IBEC, the University of Barcelona and Hospital Clinic show that, contrary to expectation, age could be a protective factor against the rapid tumor development induced by this respiratory disturbance of sleep and its immediate consequence, intermittent hypoxia.
Young at heart: novel temporal window of cardiac regeneration
A study carried out at CMR[B] in collaboration with IBEC and the UB has established that the ability of the heart to regenerate after a wound is related to the stiffness of its cellular environment and not only to the proliferative capacity of the cardiac cells, narrowing the window of regeneration to 48 hours after birth. The research, published in Science Advances, paves the way for the development of therapies based on the pharmacological modification of the extracellular matrix to promote tissue regeneration after a heart attack or stroke.

