ABOUT
The main goal of the Nanoscale Bioelectrical Characterization group is to develop a multiscale and multimodal (electrical, mechanical) approach to Bioelectricity, covering from the nano- to the microscale. To this end the group combines methods and techniques from Scanning Probe Microscopy, Artificial Intelligence and Organic Bioelectronics. The main objective is to contribute to develop new label-free characterization tools for Life Sciences, new nanomedical diagnosis approaches and new electronic biosensors.
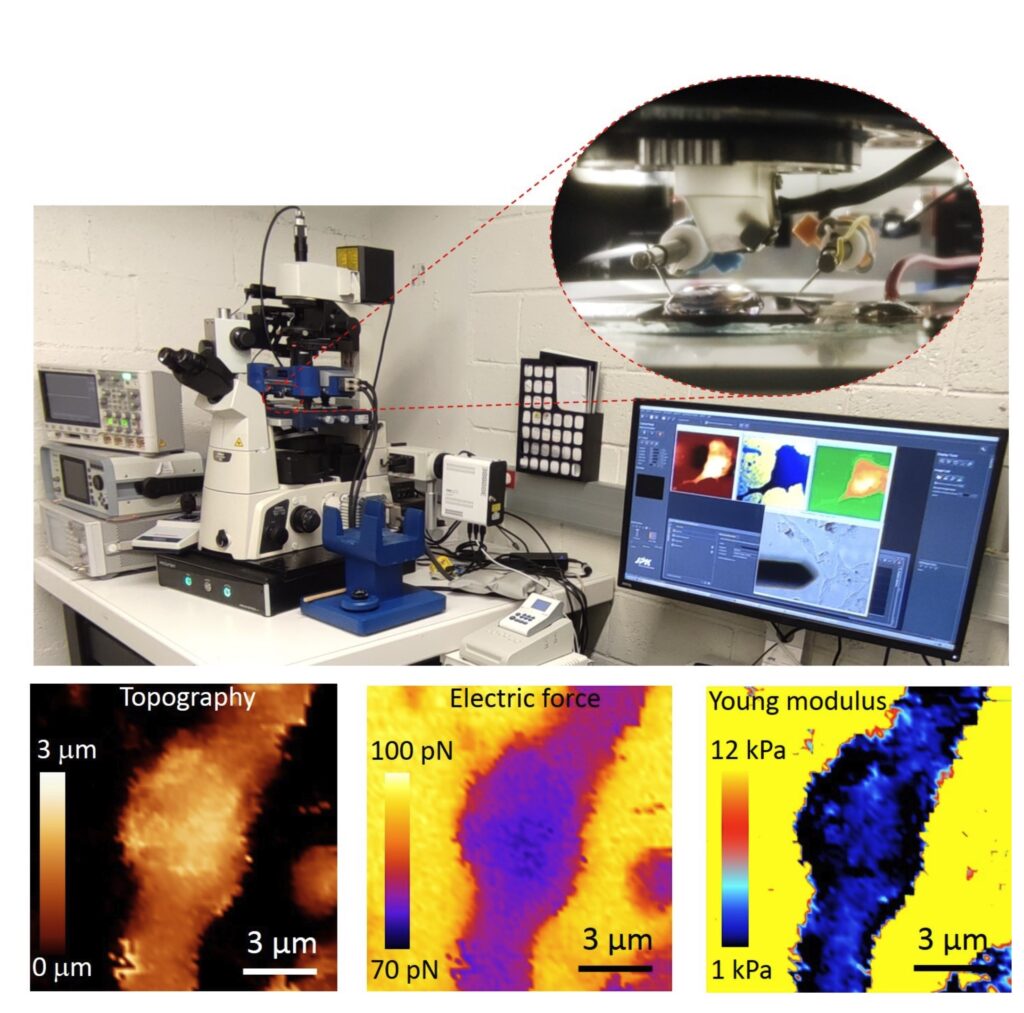
Autonomous multimodal scanning probe microscopes for Life Sciences
At present the group focuses in the development of an Autonomous Multimodal Functional Scanning Probe Microscope assisted by Artificial Intelligence for Life Sciences and Medical applications. The objective is to map the structural, electrical and mechanical properties at the nanoscale of cells, bacteria, drug nanocarriers and organic Bioelectronic devices with minimal intervention of the operator and at high throughput.
The objective is to obtain in an autonomous way fast functional electric and mechanical nanoscale maps of Life Science samples and Organic Electronics devices in physiological conditions with minimal intervention of the operator and at high throughput.
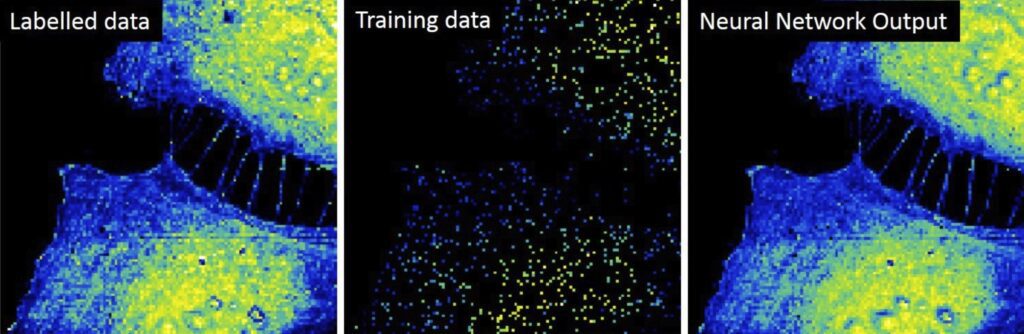
Initial results obtained by the group include the upgrade of the Scanning Dielectric Microscope to enable its operation in physiological buffers for living cell imaging, the development of a supervised machine learning algorithm to process Scanning Dielectric Microscopy data and provide almost instantaneously local dielectric constant maps of both eukaryotic and prokaryotic cells, and the implementation of a workflow for Scanning Dielectric Microscopy for high throughput and automatic nanoscale multimodal (electrical and mechanical) characterization.
High throughput multimodal characterization of drug nanocarriers
The development of novel drug nanocarriers require an exhaustive multiparametric characterization, which includes its morphology and structure, net charge, particle size distribution or phase transition temperature. These characteristics are obtained usually from different techniques. We target to obtain simultaneously and at high throughput multiparametric information on drug nanocarriers by using a single instrument, namely, the autonomous multimodal in liquid Scanning Dielectric Microscope. We aim at obtaining information on the size, sphericity, membrane wall thickness, lamellarity, Young’s modulus, stiffness, surface charge and membrane specific capacitance of drug nanocarriers, such as liposomes, polymeric nanoparticles or lipid nanoparticles.
Interrelation of mechanical and electrical processes in living neurons
Mechanical and electrical processes in cells and tissues can sometimes appear interrelated, as for instance, in the action potential propagation in neurons, which provokes the electrical polarization of the cell membrane and, at the same time, a change in neuron’s membrane tension. Similarly, the restructuring of the cytoskeleton of neurons, as occurring in the Alzheimer disease, can induce a change in cellular stiffness and, consequently, an improper neuron firing. We aim at investigating this interrelation by means of the multimodal in liquid Scanning Dielectric Microscope applied to living neurons.
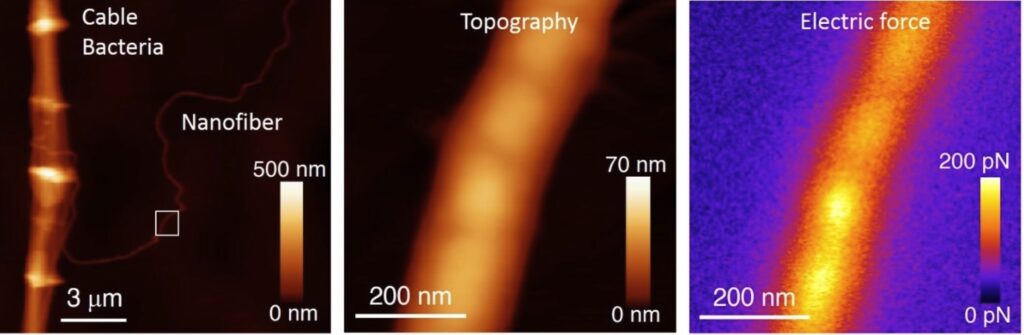
Unravelling the electrical conduction properties of cable bacteria
Long-range electron conduction in cable bacteria filaments presents unusual characteristics in the biological world, exceeding by more than 6 orders of magnitude the conductivity of the best conducting protein nanowires. Electric conduction takes place through Niquel rich protein nanofibers located in the bacteria periplasm, but still many aspects of the electronic conduction in cable bacteria remain unknown. We aim at providing new insights on the conducting properties of cable bacteria by using the unique capabilities and versatility of the Scanning Dielectric Microscope.
Novel nanoscale physical phenotyping of cancer cells
The whole process of cancer aggression, from local growth to extravasation into blood vessels, migration, seeding into different organs and formation of metastases involves physical changes (mechanical and electrical) and their interplay with protein expression and genetic transformations. We aim at developing a high throughput nanoscale multimodal physical phenotyping method for cancer cells based on the Scanning Dielectric Microscope. Our ling term objective is to provide additional diagnostics tools to medical doctors for evaluating cancer progression and aggression.
Structure-function relationships for materials in Organic Bioelectronics
Organic semiconductor materials have emerged as key materials in the development of platforms (e.g. electrolyte gated transistors) for transducing and amplifying biological and biochemical signals. This fact makes them an integral part of diverse biosensing and bioelectronic devices able to sense even single molecules or to record bioelectric potentials from excitable cells. The fundamental understanding of the nanoscale electronic and ionic transport governing the operation of these materials and devices remains, however, poorly understood. We aim at providing new insights into the structure-function relationship of organic materials used in Bioelectronics with the unique capabilities of the multimodal in operando in-liquid Scanning Dielectric Microscope.
STAFF
Staff members:
Former members:
Harishankar Balakrishnan | PhD Student
Now: Post-doc, University of Munich (Germany)
Ignacio Casuso | PhD Student
Now: Staff Scientist, INSERM (France)
Maria Chiara Biagi | PhD Student
Now: In-vivo Image Analysis Scientist, AstraZenca (Spain)
Marti Checa | PhD Student
Now: R&D Staff scientist, Oak Ridge National Laboratory (USA)
Martin Edwards | Postdoc
Now: Assistant Professor, University of Arkansas (USA)
Daniel Esteban Ferrer | PhD Student
Now: CEO, ViR S.L. (Spain)
Laura Fumagalli | Senior Researcher
Now: Reader, University of Manchester (UK)
Georg Gramse | PhD Student
Now: Group Leader, Johannes Kepler University of Linz (Austria)
Larisa Huetter | PhD Student
Now: IT consultant, Rewion (Germany)
Adrica Kyndiah | Postdoc
Now: Senior Scientist, Instituto Italiano di Tecnologia (Italy)
Helena Lozano | PhD Student
Now: Project Manager, CSIC (Spain)
Martina di Muzzio | PhD Student
Now: Engineer PMQ, Roche (Spain)
Jordi Otero | Postdoc
Now: Lecturer, Universitat de Barcelona (Spain)
Shubham Tanwar | PhD Student
Now: Post-doc, Italian Institute of Technology (Italy)
Romen Trujillo | PhD Student
Now: Associate Professor, Universitat de Barcelona (Spain)
Marc Van der Hofstadt | PhD Student
Now: Post-doc, CNRS (France)
PROJECTS
| INTERNATIONAL PROJECTS | FINANCER | PI |
|---|---|---|
| PRINGLE · Protein Based Next Generation Electronics (2022-2026) | European Commission, PathFinder Open | Gabriel Gomila |
| SPM4.0 · Autonomous Scanning Probe Microscopy for Life Sciences and Medicine powered by Artificial Intelligence | European Commission , MSCA-DN 2023 | Gabriel Gomila |
| NATIONAL PROJECTS | FINANCER | PI |
|---|---|---|
| ICREA Academia Award (2023-2027) | Catalan Institution for Research and Advanced Studies (ICREA) / Generalitat de Catalunya | Gabriel Gomila |
| Microscopio de fuerzas de barrido multiparamétrico autónomo y de alto rendimiento para aplicaciones en ciencias de la vida y medicina (BIOMEDSPM4.0) | MICIU/AEI and FEDER, UE | Gabriel Gomila |
| SGR-Grups de recerca consolidats (SGR-Cat 2021)_GRC | AGAUR / SGR | Gabriel Gomila |
| FINISHED PROJECTS | FINANCER | PI |
|---|---|---|
| SGR Grups de recerca consolidats (2017-2020) | AGAUR / SGR | Gabriel Gomila |
| SPM2.0 · Scanning probe microscopies for nanoscale fast, tomographic and composition imaging (2017-2020) | Marie Curie Skłodowska European Training Network (MSCA-ITN-ETN) | Gabriel Gomila (Project Coordinator) |
| NANOMICROWAVE · Microwave Nanotechnology for Semiconductor and Life Sciences (2013-2016) | MARIE CURIE – ITN | Gabriel Gomila |
| V-SMMART Nano · Volumetric Scanning Microwave Microscopy Analytical and Research Tool for Nanotechnology (2012-2016) | NMP – SME | Gabriel Gomila |
| AFM4NanoMed&Bio · European network on applications of Atomic Force Microscopy to Nanomedicine and Life Sciences | EU COST Action TD1002 | Gabriel Gomila (Management Committee Substitute Member) |
| BIOWIRESENSE · Plataforma universal para la detección de biomarcadores basada en nanocables bacterianos conductores (2017-2019) | MINECO, Explora Ciencia | Gabriel Gomila |
| NANOELECTOMOGRAPHY· Electrical nanotomography based on scanning probe microscopy for nanomaterials and biological samples (2014-2016) | MINECO (TEC2013-48344-C2-1-P) | Gabriel Gomila |
| NANOELECTROPHYS · Scanning Electric Force Microscope for Electrophysological Recordings at the Nanoscale (2016-2019) | MINECO (TEC2016-79156-P) | Gabriel Gomila |
| ICREA Academia Award (2015-2019) | Catalan Institution for Research and Advanced Studies (ICREA) / Generalitat de Catalunya | Gabriel Gomila |
| BORGES · Biosensing with ORGanic ElectronicS (2019-2022) | Marie Curie Skłodowska European Training Network (MSCA-ITN-ETN) | Gabriel Gomila |
| BIGDATASPM · Métodos de datos masivos aplicados a la Microscopía de Sonda de Barrido para estudios eléctricos funcionales en ciencias de la vida (2020-2023) | MINECO, Generación Conocimiento: Proyectos I+D | Gabriel Gomila |
| Correlative Electrical and Mechanical Scanning Probe Microscopy for Life Science Application | Beatriu de Pinós 2019/ AGAUR | Aurora Dols |
PUBLICATIONS
Check for more detailed information on the outputs of the Group at IBEC CRIS portal.
Publications list:
EQUIPMENT
- Cypher Atomic Force Microscope (Asylum Research)
- Nanowizard 4 Bio-Atomic Force Microscope (JPK)
- Cervantes Atomic Force Microscope (Nanotec Electronica)
- Easy Scan 2 Atomic Force Microscope (Nanosurf)
- AxioImager A1m Reflection Optical Microscope (Zeiss) equipped with a AxioCam ERc5s (Zeiss)
- CompactStat portable electrochemical interface and impedance analyzer (Ivium Technologies)
- Palmsens 4, 8 channel Potentiostat (Palmens)
- 2 eLockIn204 4-phase Lock-In amplifiers (Anfatec)
- Keithley 6430 sub-femtoAmp remote sourcemeter
- Keysight B2912A precision Source/Measure Unit, 2 channels
- Keysight N9310A RF Signal Generator 9 kHz to 3.0 GHz
- Computation Workstation Intel Xeon, NVIDIA RTXA5000
COLLABORATIONS
- Dr. Filip Meysman
University of Antwerp, Belgium - Dra. Adrica Kyndiah
Italian Institute of Technology, Italy - Dr. Martí Checa
Oak Ridge National Laboratory, USA - Dr. Jordi Borrell
University of Barcelona, Spain - Dra. Marta Mas-Torrents
Institut de Ciències de Materials de Barcelona, Spain - Dr. Eduard Torrents
Institut de Bioenginyeria de Catalunya, Spain - Dr. Jose Antonio del Rio
Institut de Bioenginyeria de Catalunya, Spain
NEWS

New insights into how bacteria control DNA synthesis open the door to next‑generation antimicrobials
A study led by the Institute for Bioengineering of Catalonia (IBEC) and the Molecular Biology Institute of Barcelona (IBMB) provides the most detailed picture to date of NrdR — the master regulator of ribonucleotide reductases (RNRs) in bacteria. Researchers obtained the first detailed images of the complete NrdR protein structure and showed how changes in the shape and grouping of this protein affect the way it controls key processes inside the cell. The findings, recently published in International Journal of Biological Macromolecules, increase our understanding of how bacteria regulate the production of the molecular building blocks of DNA, a crucial aspect for both fundamental microbiology and the development of new antimicrobial strategies.

Berlin Hosts the Mid-Term Meeting and First Workshop of the SPM4.0 Network coordinated by IBEC
The SPM4.0 consortium convened at Charité–Berlin for its first training workshop and mid-term meeting, reinforcing scientific collaboration and supporting the development of the project’s doctoral researchers. The sessions offered a comprehensive overview of ongoing scientific progress and future training activities. This milestone moment further strengthened coordination across the network and set the pace for upcoming project objectives.

IBEC hosts the EU Project SPM4.0 kick-off meeting
Researchers from across Europe gathered for the kick-off meeting of the SPM4.0 project, an innovative Marie Curie Skłodowska Doctoral Network (MSCA-DN) dedicated to advancing the capabilities of autonomous Scanning Probe … Read more

IBEC and VHIR hold a collaboration day to promote synergies
The 1st Translational Collaboration Day between the Vall d’Hebron Institute of Research (VHIR) and the Institute of Bioengineering of Catalonia (IBEC), held on 21st November, was an opportunity to learn about the projects and research lines of both institutions and to promote interaction between professionals.

IBEC and BST strengthen ties with Translational Collaboration Day
IBEC and the Blood and Tissue Bank of Catalonia (BST) held a day to explore new collaborations in bioengineering and translational medicine. The meeting, held yesterday at IBEC, highlighted innovative projects, presented a joint PhD programme and strengthened the link between biomedical research and clinical applications.

Bioengineering for Emergent and Advanced Therapies at the 17th IBEC Symposium
IBEC’s 17th Annual Symposium focused on ‘Bioengineering for Emergent and Advanced Therapies’, one of IBEC’s key application areas. Around 300 people attended the event, including local and international researchers. It was a multidisciplinary environment in which experts from other centres and the IBEC community itself had the opportunity to present their projects and share knowledge.

Two projects with IBEC participation selected in the MSCA call for PhD networks
IBEC will coordinate SPM4.0 and participate as a partner in ENTRY-DM, two of the projects selected in the 2023 call for PhD networks within the Marie Skłodowska-Curie Actions (MSCA). Thanks to these two projects, IBEC will add three new PhD students to its staff.

New Methodology for Studying Organic Transistors in Operation with Applications in Bioelectronics
A study led by IBEC has successfully mapped the local electrical potential along the structure of organic transistors used in bioelectronics, enabling a detailed assessment of bottlenecks in charge transport. The goal of this study is to deepen the understanding of the properties of charge transport in materials used in organic electronics when in contact with liquid media, aiming to enhance their applications in biosensors or bioelectrical recordings.

Elisabeth Engel and Gabriel Gomila receive the ICREA Acadèmia grant
IBEC researchers Elisabeth Engel and Gabriel Gomila have been awarded the “ICREA Acadèmia” distinction by the Catalan Institution for Research and Advanced Studies (ICREA). Engel and Gomila, who lead their … Read more

Research Assistant at the Nanoscale bioelectrical characterization group
Introduction to the vacant position: The Nanobioelec Group/Unit is looking for Research Assistant. The contract will be within the framework of the European Project PRINGLE, whose objective is to develop … Read more
JOBS

Senior Researcher position at the Nanoscale Bioelectrical Characterization research group
Ref: SR_GG // Deadline: 08/12/2024

Senior Laboratory Technician at the Nanoscale Bioelectric Characterization Research Group (Ref SRT_GG)
Ref: SRT_GG // Deadline: 20/06/2024

Post-doctoral researcher at the Nanobioelec Research Group (PD_GG)
Ref: PD_GG // Deadline: 15/01/2024

Predoctoral researcher at the Nanoscale Bioelectrical Characterization Research Group
Ref: FPI_GG /Deadline: 31/10/2023

Post-doctoral researcher at the Nanobioelec Research Group (PD_GG)
Ref: PD_GG // Deadline: 21/07/2023

Pre-doctoral researcher at the Nanobioelec Research Group (Phd_GG)
Ref: Phd_GG // Deadline 30/06/2023

Post-doctoral researcher at the Nanobioelec Research Group (Pd_GG)
Ref: Pd_GG // Deadline: 26/11/2023

Predoctoral researcher at the Nanoscale bioelectrical characterization group (PHD_GG)
Reference: PhD-GG / Deadline: 15/03/2023


 ibecbarcelona.eu
ibecbarcelona.eu