Researchers at the Institute for Bioengineering for Catalonia (IBEC), the Polytechnic University of Catalonia – BarcelonaTech (UPC) and the International Centre for Numerical Methods in Engineering (CIMNE) have developed a new strategy for ‘programming’ the shape of biological tissues in vitro. The study, published in the journal Science, is the first to demonstrate that it is possible to guide the forces and final shape of living tissue by controlling the orientation of its cells. The research opens the door to new applications in tissue engineering, biohybrid robotics, and the design of smart living materials.

Biological tissues have a remarkable ability to organise and change shape, driven by forces generated by their own cells. One of the major challenges in bioengineering is harnessing this natural behaviour to design synthetic living materials capable of adopting predetermined shapes. However, precisely controlling how a tissue behaves and directing its internal forces to adopt the desired shape remains a significant scientific challenge.
We are demonstrating that we can design the shape a living tissue will adopt just by by controlling how its cells are oriented.
Xavier Trepat
A new study led by the Institute for Bioengineering of Catalonia (IBEC), the Polytechnic University of Catalonia – BarcelonaTech (UPC) and the International Centre for Numerical Methods in Engineering (CIMNE) in collaboration with the European Molecular Biology Laboratory (EMBL) in Barcelona, presents a strategy for ‘programming’ these shape changes by controlling how cells orient themselves within the tissue through chemical patterns.
The result is living tissue that can deform in a controlled manner to generate reproducible three-dimensional structures. This is particularly significant for Xavier Trepat, ICREA Research Professor at IBEC, leader of the Integrative Cell and Tissue Dynamics group, and co-lead author of the study:
“We are demonstrating that we can design the shape a living tissue will adopt just by by controlling how its cells are oriented.”, emphasises Trepat, who is also a professor at the University of Barcelona (UB) and a member of the Centre for Biomedical Research Network in Bioengineering, Biomaterials and Nanomedicine (CIBER-BBN).
The scientists present the study’s results as a means of designing living surfaces that can change shape independently, with potential applications ranging from tissue engineering to biohybrid robotics.
Cells that align and shape tissues
Biological tissues composed of elongated cells tend to self-organise into multicellular domains, with all the cells aligned in the same direction — much like the fibres of a textile. This is known as nematic order.
Occasionally, however, nematic order breaks down at specific points, known as topological defects. These are locally disordered areas comparable to the whirls or bifurcations found in a fingerprint. In biology, these defects act as points where forces converge and can influence how tissues grow, migrate or deform.
‘The orientation of the cells controls the forces, and the forces can control the generation of a three-dimensional shape,’ explains Pau Guillamat, a researcher in the Integrative Cell and Tissue Dynamics group at IBEC and the study’s first author.
To direct these forces, the team used chemical micropatterns to ‘draw’ lines on flat surfaces. They did this by using a protein to which the cells adhere, and then surrounded these lines with areas containing a polymer to which the cells do not adhere. Thanks to these patterns, the cells aligned themselves along the lines, creating the desired ‘map’ of orientations.
This enabled topological defects to be imposed in specific locations — something that nature generates spontaneously, albeit in a disorderly manner. “The key is that we can decide where these defects will be, and therefore where the forces will be generated within the tissue,” says Guillamat.
The crucial experiment came when the researchers peeled the tissue away from the substrate on which the cells were growing. While it was still attached, the internal forces generated by the cells were anchored to the support, preventing the tissue from changing shape. However, by removing this mechanical constraint, the accumulated stress was able to redistribute itself freely.
“It’s like an elastic sheet that is stretched tight and fixed at the edges. While it is held in place, it does not deform; however, when released, it adopts a new geometry determined by the internal stresses,” explains Guillamat.
The same thing happens with cellular tissue: when peeled away, it contracts and deforms rapidly in the direction of the stress created by the cells’ orientation and topological defects.
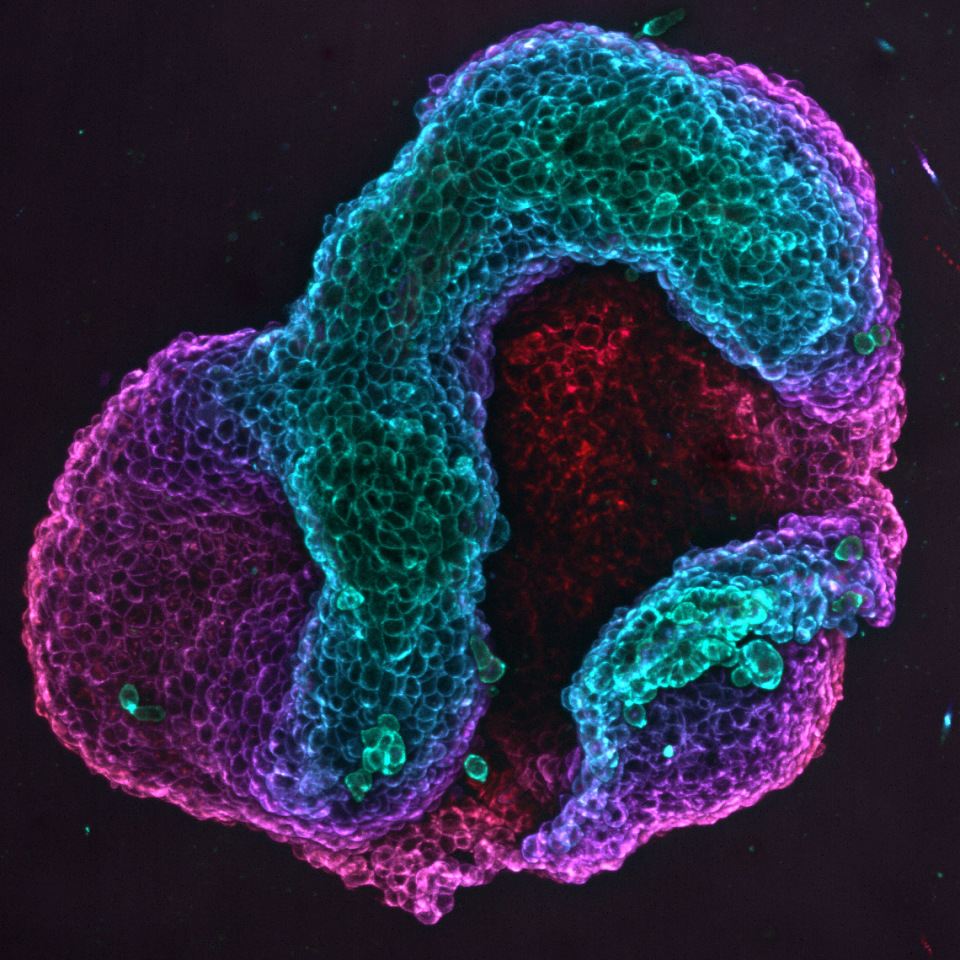
Simulations to predict the final shape
To gain a deeper understanding of the origin of tissue forces and shapes, and to predict them, the team led by Marino Arroyo — full professor in the Department of Civil and Environmental Engineering at the UPC, principal investigator of the Soft and Living Material Interfaces research group at CIMNE and co-lead author of the study — developed theoretical models and simulations. These models and simulations enable the team to anticipate how a specific pattern of cell orientations will eventually transform into a specific three-dimensional shape.
According to Arroyo, ‘Our models have allowed us to examine different hypotheses and ultimately identify the mechanism by which cell orientation leads to the three-dimensional folding of tissues. Furthermore, they provide a quantitative relationship between the nematic pattern and the shape’, thus confirming that the system can be used as a predictive platform for the morphological design of tissues.
A wide range of applications
These systems can be considered living materials that generate programmable forces and shapes and can also integrate information and respond intelligently.
Pau Guillamat
This proof-of-concept research opens the door to many applications, such as tissue engineering to create three-dimensional structures without the need for artificial scaffolds, biohybrid robotics using deformable living tissues as biological actuators, and the design of smart living materials — living surfaces capable of reconfiguring their shape and potentially their functional properties.
“These systems can be considered living materials that generate programmable forces and shapes and can also integrate information and respond intelligently,” adds Guillamat.
In addition to its potential applications, this methodology enables the study of biological phenomena such as organ formation and tumour behaviour. ‘It is a perfect tool for understanding how patterns of cell orientation influence the mechanics and evolution of complex tissues,’ says Trepat.
The study also involved the collaboration of the IBEC’s Cellular and Molecular Mechanobiology group, led by Pere Roca-Cusachs, who is also a full Professor at the Faculty of Medicine and Health Sciences of the University of Barcelona (UB).
Referenced paper
Pau Guillamat, Waleed Mirza, Pradeep K. Bal, Manuel Gómez-González, Pere Roca-Cusachs, Marino Arroyo, Xavier Trepat.Guidance of cellular nematic elastomers into shape-programmable living surfaces. Science (2026). DOI: 10.1126/science.adz9174





