In an international collaboration, researchers led by Nuria Montserrat, ICREA Research Professor at IBEC, have generated human mini-kidneys that simulate the kidney of a person with diabetes in the early stages of the disease. These diabetic mini-kidneys open the door to studying, among others, the relationship between diabetes and COVID-19.
For two years, thousands of scientists and doctors around the world have been working to understand how COVID-19 develops and what relationship it has with other types of diseases. Various studies indicate that people with diabetes are more likely to develop severe COVID-19, as well as that more than 20% of patients hospitalized for COVID-19 suffer acute kidney damage. However, to date, it was unknown what was the factor that caused this to happen.

Now, an international team led by Nuria Montserrat, ICREA research professor at the Institute of Bioengineering of Catalonia (IBEC) and principal investigator of the “Pluripotency for organ regeneration” group, in collaboration with, among others, researchers from the University of Florida, the Life Sciences Institute of the University of British Columbia in Canada, Karolinska Institutet and Karolinska University Hospital in Sweden have used bioengineering to develop mini-kidneys that simulate the kidney of a person in the early stages of diabetes.
Our diabetic renal organoid model has allowed us to observe that diabetic mini-kidneys, with a greater number of ACE2 receptors, have a greater susceptibility to viral infection.
Elena Garreta, Institute for Bioengineering of Catalonia and first co-author of the study
In this international collaboration the researchers have provided, for the first time, the use of kidney organoids to understand the early stages of diabetes in this organ. In order to demonstrate that the ACE2 receptor plays an essential role in SARS-CoV-2 infection in the kidney, the team has also used genetic engineering to generate defective organoids for other receptors described to date as gateways for the virus. Using patient kidney cells, this study also reveals the role of energy metabolism in SARS-CoV-2 infection, opening the door to the identification of new therapeutic interventions to treat COVID-19. This groundbreaking study has just been published in the prestigious journal Cell Metabolism.
Diabetic mini-kidneys have more portals of entry for SARS-CoV-2
It is absolutely imperative to understand the molecular mechanisms that underlie more severe COVID-19 in patients with diabetes and other metabolic comorbidities. The development of a diabetic kidney organoid is a great step towards experimentally dissecting how metabolic changes can impact SARS-CoV-2 infections. The data again demonstrate that ACE2 is the essential receptor for SARS-CoV-2 even under conditions of comorbidity.
Josef Penninger, Institute of Molecular Biotechnology of the Austrian Academy of Sciences
Mini-kidneys have been developed in the laboratory from pluripotent human stem cells. To reproduce the diabetic environment, the researchers have subjected the mini-kidneys to culture conditions that result in the generation of mini-kidneys with the same cellular characteristics and metabolic alterations as those found in the kidneys of a person with early-stage diabetes.
Using different molecular biology techniques, such as gene editing, the researchers have observed that, in diabetic mini-kidneys, it is the abundance of the ACE2 receptor that determines susceptibility to viral infection, establishing a causal relationship between diabetes and the presence of one of the receptors described so far as determinant in SARS-CoV-2 infection.
Furthermore, using state-of-the-art techniques such as RNA sequencing, the researchers identified that diabetic mini-kidneys have a metabolic signature that could explain why diabetic mini-kidneys become more infected.
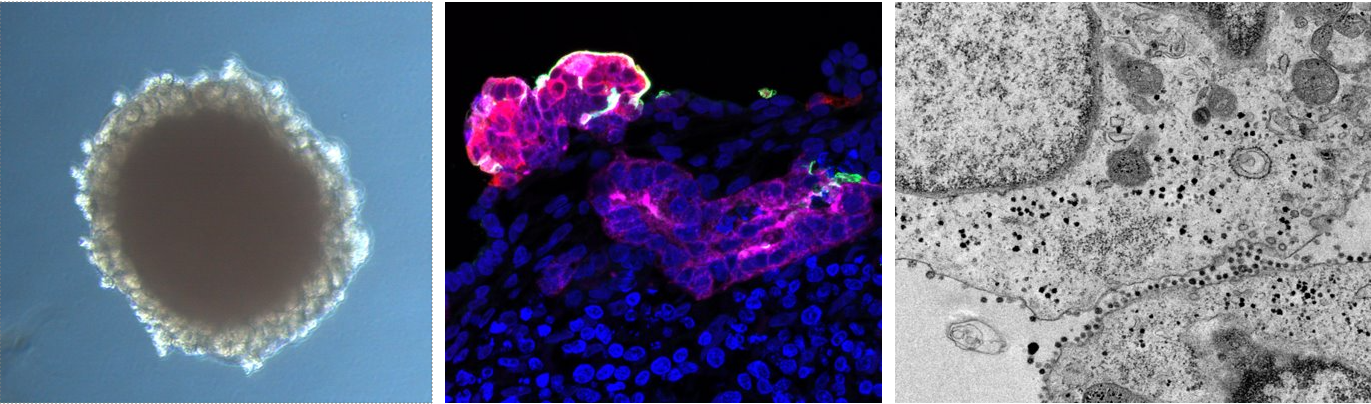
Bright field image of a kidney organoid generated from human pluripotent stem cells [left image]. A high magnification detail of a a proximal tubular structure (magenta) within a kidney organoid after SARS-CoV-2 infection, that shows ACE2 positive cells (green) and SARS-CoV-2 infected cells (red) [middle image]. Transmission electron microscopy image showing a detail of kidney organoid cells infected with SARS-CoV-2 [right image].
Diabetes increases susceptibility to SARS-CoV-2 infection in patient cells
This finding sheds a light on a potential mechanism behind more severe cases of diabetic patients. This technology will improve our capability to investigate how the virus interacts with different organs in the human body.
Ali Mirazimi, adjunct professor at Karolinska Institutet and one of the study’s corresponding authors.
To verify whether the results obtained with the mini-kidneys are also observed in the native organ, the researchers analyzed kidney cells from patients with diabetes and individuals without diabetes. The data show that kidney cells from diabetic patients, in the same way as what happens in mini-kidneys, have more ACE2 receptors and suffer a higher rate of SARS-CoV-2 infection. To delve into the mechanisms that may explain such observations, the researchers used a compound that modulates the metabolic state of cells and found that the treatment reduced viral infection.
We have shown that the SARS-CoV-2 virus is capable of directly infecting proximal tubule cells isolated from the human kidney and that diabetes makes these cells more prone to infection.
Megan Stanifer, first co-author of the study (University of Florida)
This research was supported in part by the Institute of Health Carlos III and the Foundation Banco Bilbao Vizcaya through emergency funding focused on accelerating the development, testing, and implementation of measures to deal with the COVID-19 outbreak.
Nuria Montserrat is a member of the Biomedical Research Center – Bioengineering, Biomaterials and Nanomedicine Network (CIBER-BBN).
Reference article: Elena Garreta, Patricia Prado, Megan Stanifer, Vanessa Monteil, Andrés Marco, Asier Ullate-Agote, Daniel Moya-Rull, Amaia Vilas-Zornoza, Carolina Tarantino, Juan Pablo Romero, Gustav Jonsson, Roger Oria, Alexandra Leopoldi, Astrid Hagelkruys, Maria Gallo, Federico González, Pere Domingo-Pedrol, Aleix Gavaldà, Carmen Hurtado del Pozo, Omar Hasan Ali, Pedro Ventura-Aguiar, Josep María Campistol, Felipe Prosper, Ali Mirazimi, Steeve Boulant, Josef M. Penninger, Nuria Montserrat. A diabetic milieu increases ACE2 expression and cellular susceptibility to SARS-CoV-2 infections in human kidney organoids and patient cells. Cell Metabolism. 2022.





