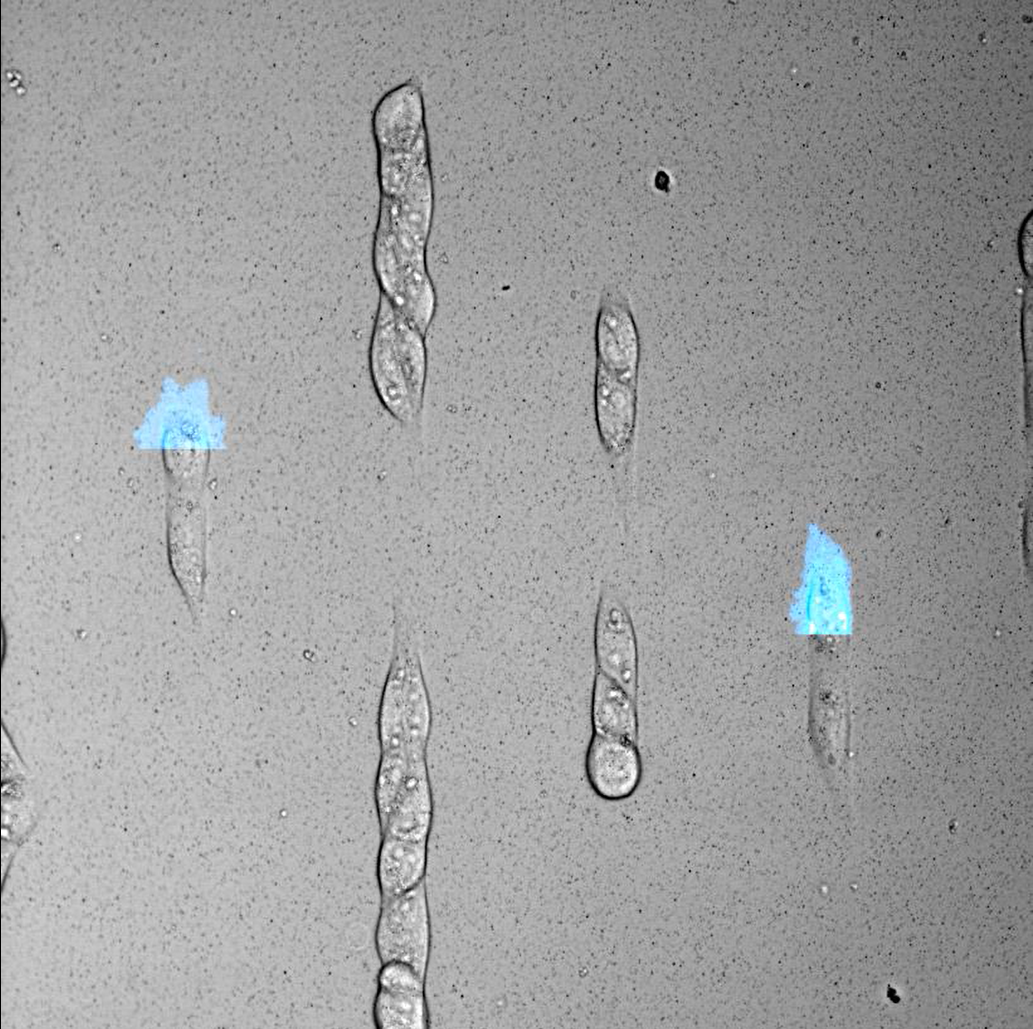
Scientists create leader cells with light
A study led by the Institute for Bioengineering of Catalonia (IBEC) has studied the migratory movement of groups of cells using light control. The results show that there is no leader cell that directs the collective movement, as previously thought, but that all cells participate in the process. These findings are relevant to the design of treatments to stop tumour invasion or accelerate wound healing, physiological processes closely linked to cell migration.